Preparation of Sinomenine Microemulsion by D-Optimal Mixture Design and Evaluation of Its Pharmacodynamics
-
摘要:
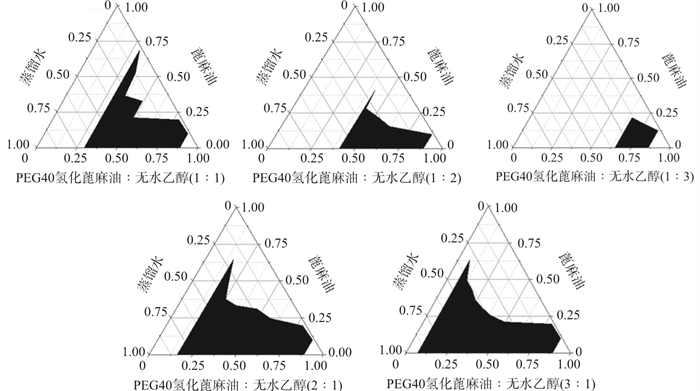
目的 优化青藤碱微乳的制备工艺, 进行初步药效学评价。 方法 建立青藤碱的HPLC含量测定方法, 通过溶解度实验及伪三元相图等初步筛选青藤碱微乳处方, 利用D-最优混料实验设计法以粒径和载药量为考察指标进行青藤碱微乳处方优化, 并对其粒径、载药量及稳定性等进行评价; 采用体外透皮实验考察经皮吸收情况, 进行耳肿胀实验评价抗炎药效。 结果 以甲醇∶0.1%磷酸(40∶60)为流动相, 检测波长为262 nm测定青藤碱含量, 方法适用性好; 获得微乳最佳处方为蓖麻油占比7.0%, PEG40氢化蓖麻油/无水乙醇为混合乳化剂占比69.0%, 最佳Km值为3∶1, 蒸馏水占比24.0%;制得微乳平均粒径18.76 nm, PDI为0.072, 载药量5.225%;1.0%青藤碱微乳12 h累积渗透量为1.223 4 μg·cm-2, 稳态渗透速率为0.064 9 μg·cm-2·h-1, 均优于青藤碱溶液; 高浓度的青藤碱微乳对小鼠耳肿胀抑制率为65.07%, 与阳性药地塞米松效果相近。 结论 制得青藤碱微乳工艺稳定, 载药量高, 透皮吸收及抗炎效果较好, 为青藤碱经皮给药制剂开发提供参考。 Abstract:OBJECTIVE To optimize the preparation process of sinomenine microemulsion and evaluate its pharmacodynamics. METHODS HPLC method for sinomenine content determination was established, and sinomenine microemulsion prescription was initially screened by solubility test and pseudo-ternary phase diagram. D-optimal mixing experimental design method was used to optimize sinomenine microemulsion prescription with particle size and drug load as investigation indexes, and its particle size, drug load and stability were evaluated. Transdermal absorption was investigated by transdermal test in vitro, and the anti-inflammatory effect was evaluated by ear swelling test. RESULTS With methanol:0.1% phosphoric acid (40∶60) as the mobile phase, the detection wavelength was 262 nm, and the method was suitable for the determination of sinomenine. The optimal formula of microemulsion was obtained as castor oil (7.0%), PEG40 hydrogenated castor oil/anhydrous ethanol (69.0%), the optimal Km value was 3∶1, and distilled water (24.0%). The average particle size of the microemulsion was 18.76 nm, the PDI was 0.072 and the drug loading was 5.225%. The cumulative permeability of 1.0% sinomenine microemulsion at 12 h was 1.223 4 μg·cm-2, and the steady permeability rate was 0.0649 μg·cm-2·h-1, which was better than sinomenine solution. The inhibitory rate of sinomenine microemulsion was 65.07%, which was similar to dexamethasone. CONCLUSION The preparation of sinomenine microemulsion has the advantages of stable process, high drug loading, good transdermal absorption and anti-inflammatory effect, which can provide reference for the development of sinomenine transdermal drug delivery preparation. -
Key words:
- sinomenine /
- microemulsion /
- pseudo-ternary phase diagram /
- d-optimal mixture design /
- pharmacodynamics
-
表 1 青藤碱在各辅料中溶解度
Table 1. Solubility of sinomenine in excipients
微乳组成 种类 溶解度/(mg·g-1) 油相 蓖麻油 0.450 4 油酸乙酯 0.003 8 油酸 0.059 7 乳化剂 聚山梨酯80 1.109 7 聚山梨酯20 1.075 7 PEG40氢化蓖麻油 1.815 4 助乳化剂 无水乙醇 3.477 5 1, 2-丙二醇 2.928 7 表 2 乳化等级分类
Table 2. Emulsification grade classification
乳化等级 外观描述 A 快速乳化, 外观呈澄清或淡蓝色乳光 B 快速乳化, 外观呈半透明或蓝白色 C 较快乳化, 外观呈亮白色不透明液体 D 乳化较慢, 外观呈暗灰色或上部略带油滴 E 不发生乳化, 上部有油滴 表 3 青藤碱微乳辅料配伍实验结果
Table 3. Experimental results of sinomenine microemulsion excipient compatibility
油相 乳化剂 助乳化剂 现象 放置 3d 后现象 油酸 聚山梨酯80 无水乙醇 C C 蓖麻油 聚山梨酯80 无水乙醇 E E 油酸 聚山梨酯80 1, 2-丙二醇 C~D D 蓖麻油 聚山梨酯80 1, 2-丙二醇 D~E D-E 油酸 PEG40氢化蓖麻油 无水乙醇 A~B B 蓖麻油 PEG40氢化蓖麻油 无水乙醇 A A 油酸 PEG40氢化蓖麻油 1, 2-丙二醇 B B~C 蓖麻油 PEG40氢化蓖麻油 1, 2-丙二醇 B~C A~B 表 4 D-最优混料设计及结果(x±s,n=3)
Table 4. D-optimal mixture design and results(x±s,n=3)
序号 X1/% X2/% X3/% Y/nm Z/% 综合评分M 1 28.5 31.5 40.0 115.25 6.128 140 2 43.3 26.1 30.6 137.50 4.007 100 3 7.0 69.0 24.0 18.86 5.225 170 4 69.5 10.0 20.5 180.73 0.040 30 5 62.8 26.1 11.1 192.03 0.143 20 6 7.0 53.0 40.0 125.25 4.459 100 7 89.0 10.0 1.0 122.83 0.114 50 8 7.0 61.0 32.0 28.24 3.460 140 9 21.8 47.6 30.6 78.13 1.418 90 10 50.0 10.0 40.0 95.38 7.446 160 11 36.6 41.2 21.2 115.10 2.350 90 12 30.0 69.0 1.0 54.06 3.056 130 13 21.8 55.6 22.6 38.28 5.241 160 14 18.5 69.0 12.5 40.55 2.510 130 15 33.3 55.6 11.1 56.95 4.403 140 16 59.5 39.5 1.0 147.90 0.591 40 表 5 青藤碱各体系体外透皮吸收的比较(n=3)
Table 5. Comparison of transdermal absorption of sinomenine systems in vitro (n=3)
体系 回归方程 R2 Q12/(μg·cm-2) Js/(μg·cm-2·h-1) 1%青藤碱水溶液 Q=0.046 0t+0.4516 0.983 2 1.003 1±0.004 8 0.046 0 1%青藤碱微乳 Q=0.064 9t+0.450 9 0.995 5 1.223 4±0.019 5 0.064 9 表 6 青藤碱微乳对二甲苯所致小鼠耳肿胀的影响(x±s,n=8)
Table 6. Effect of sinomenine microemulsion on xylene induced ear swelling in mice(x±s,n=8)
组别 剂量/(g·kg-1) 肿胀度/mg 肿胀率/% 抑制率/% 空白组 — 0 0 — 模型组 — 43.8±1.8 99.54 — 地塞米松组 0.005 16.6±1.3** 25.70 74.18 低浓度微乳组 0.500 40.0±2.6** 85.65 13.95 高浓度微乳组 2.000 24.2±1.1** 34.77 65.07 注: 与模型组比较,*P < 0.05, **P < 0.01。 -
[1] 吴红根, 仇萍, 彭晓珊, 等. 不同盐酸青藤碱制剂的毛细管电泳分析方法[J]. 中成药, 2021, 43(4): 1100-1102. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYA202104050.htmWU H G, QIU P, PENG X S, et al. Capillary electrophoresis analysis of different sinomenine hydrochloride preparations[J]. Chin Tradit Pat Med, 2021, 43(4): 1100-1102. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYA202104050.htm [2] 王玺, 张智勇, 仇萍, 等. 青风藤、青藤碱及其相关制剂的研究进展[J]. 中国药学杂志, 2021, 56(2): 85-93. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYX202102001.htmWANG X, ZHANG Z Y, QIU P, et al. Research progress on caulis sinomenii, sinomenine and related preparations[J]. Chin Pharm J, 2021, 56(2): 85-93. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGYX202102001.htm [3] 丁杨, 钱珊珊, 郭健, 等. 基于双位点同步微透析技术的青藤碱微乳凝胶皮肤及血液药代动力学研究[J]. 南京中医药大学学报, 2022, 38(11): 1050-1055. doi: 10.14148/j.issn.1672-0482.2022.1050DING Y, QIAN S S, GUO J, et al. Pharmacokinetic study of skin and blood of sinomenine-loaded microemulsion gel based on double sites simultaneous microdialysis[J]. J Nanjing Univ Tradit Chin Med, 2022, 38(11): 1050-1055. doi: 10.14148/j.issn.1672-0482.2022.1050 [4] 黎绫, 龙爱平, 王碧霞, 等. 微乳给药系统的研究与应用进展[J]. 湘南学院学报(医学版), 2020, 22(3): 69-71. https://www.cnki.com.cn/Article/CJFDTOTAL-CZYG202003025.htmLI L, LONG A P, WANG B X, et al. Research and application progress of microemulsion drug delivery system[J]. J Xiangnan Univ Med Sci, 2020, 22(3): 69-71. https://www.cnki.com.cn/Article/CJFDTOTAL-CZYG202003025.htm [5] 刘筱雅, 江昌照, 高文彦, 等. 微乳和基于微乳的经皮给药制剂的研究进展[J]. 中国医药工业杂志, 2020, 51(4): 442-449. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHOU202004004.htmLIU X Y, JIANG C Z, GAO W Y, et al. Progress of microemulsion and microemulsion-based transdermal preparations[J]. Chin J Pharm, 2020, 51(4): 442-449. https://www.cnki.com.cn/Article/CJFDTOTAL-ZHOU202004004.htm [6] 闫巧, 薛玉叶, 李方琴, 等. 甘草酸新型离子液体微乳的制备及体外评价[J]. 中草药, 2023, 54(1): 62-71. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202301009.htmYAN Q, XUE Y Y, LI F Q, et al. Preparation and in vitro evaluation of a novel ionic liquid microemulsion of glycyrrhizic acid[J]. Chin Tradit Herb Drugs, 2023, 54(1): 62-71. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202301009.htm [7] 马书伟, 王永洁, 陈柽, 等. D-最优混料设计优化复方甘草微乳制备工艺的研究[J]. 中国中药杂志, 2018, 43(6): 1131-1138. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201806009.htmMA S W, WANG Y J, CHEN C, et al. Optimize preparation of compound licorice microemulsion with D-optimal design[J]. China J Chin Mater Med, 2018, 43(6): 1131-1138. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201806009.htm [8] 陆姗姗, 陈军, 赵玉荣, 等. 小茴香油微乳凝胶的制备及其体外透皮促渗行为研究[J]. 中国现代应用药学, 2022, 39(8): 1067-1074. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYD202208010.htmLU S S, CHEN J, ZHAO Y R, et al. Research on preparation of fennel oil microemulsion and promotion of transdermal permeability in vitro[J]. Chin J Mod Appl Pharm, 2022, 39(8): 1067-1074. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYD202208010.htm [9] 郭雪, 陈金素, 许蕊蕊, 等. 基于"药辅合一" 的桉叶油乳膏的制备及药效研究[J]. 中国现代中药, 2022, 24(4): 576-582. https://www.cnki.com.cn/Article/CJFDTOTAL-YJXX202204002.htmGUO X, CHEN J S, XU R R, et al. Preparation and pharmacodynamic study of Eucalyptus oil cream based on "unification of medicine and adjuvant"[J]. Mod Chin Med, 2022, 24(4): 576-582. https://www.cnki.com.cn/Article/CJFDTOTAL-YJXX202204002.htm [10] 陈云艳, 马睿, 卜晓阳, 等. 基于"药辅合一" 的紫杉醇-薏苡仁油微乳的制备及含量测定[J]. 中国现代应用药学, 2023, 40(8): 1064-1069. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYD202308007.htmCHEN Y Y, MA R, BU X Y, et al. Preparation and detemination of paclitaxel-Coix seed oil microemulsion based on the unity of medicine and adjuvant[J]. Chin J Mod Appl Pharm, 2023, 40(8): 1064-1069. https://www.cnki.com.cn/Article/CJFDTOTAL-XDYD202308007.htm [11] 张宇航, 高寒, 徐伟, 等. D-最优混料设计法优化刺五加总苷微乳制备工艺及肠吸收特性研究[J]. 中国中药杂志, 2022, 47(12): 3233-3241. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202212014.htmZHANG Y H, GAO H, XU W, et al. Optimization of preparation technology for Acanthopanax senticosus total saponins microemulsion by D-optimal mixture design and intestinal absorption characteristics[J]. China J Chin Mater Med, 2022, 47(12): 3233-3241. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202212014.htm [12] 王景雁, 马书伟, 赵馨雨, 等. 复方甘草微乳凝胶剂的制备与药效学评价[J]. 中国中药杂志, 2020, 45(21):5193-5199. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202021015.htmWANG J Y, MA S W, ZHAO X Y, et al. Preparation of compound liquorice microemulsion gel and its pharmacodynamics evaluation[J]. China J Chin Mater Med, 2020, 45(21):5193-5199. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202021015.htm -





 下载:
下载: