Study on the Characteristic Specturm and Color Change of Steamed Rehmanniae Radix Praeparata and Optimization of Processing Technology
-
摘要:
目的 研究熟地黄蒸制过程中特征图谱与颜色的变化规律, 结合Box-Behnken响应曲面模型, 优选熟地黄炮制工艺。 方法 应用Pearson相关性分析、聚类分析、主成分分析(PCA)、正交偏最小二乘分析(OPLS-DA)等多元统计方法研究熟地黄蒸制过程中的颜色和特征图谱峰面积变化, 筛选熟地黄药材关键的质量标志物(Q-markers), 以Q-markers为评价指标, 结合综合评分法考察蒸制时间、干燥温度、干燥时间对熟地黄质量的影响, 同时采用Box-Behnken响应曲面法优选最佳炮制工艺。 结果 建立的生地黄特征图谱和熟地黄特征图谱分别标定18、20个共有峰, 其中19号峰[5-羟甲基糠醛(5-HMF)]为生地黄经炮制后产生的新化学成分; 生地黄蒸制2 h后色差显著(ΔE*>12), 且各特征峰峰面积与生品的差异较大; 蒸制时间超过2 h后, 不同蒸制时间点的熟地黄ΔE*为0.28~4.76, 色差不能被肉眼识别。以色度值L*、a*、b*、E*为变量可将不同炮制点的熟地黄聚为3类, 以色度值L*、a*、b*、E*, 环烯醚萜苷类成分, 苯乙醇苷类成分, 5-HMF为变量可将不同炮制点的熟地黄聚为4类。熟地黄最佳炮制工艺为: 润制水量为药材量的0.3倍, 润制时间为24 h, 蒸制时间为2.17 h, 干燥温度为61.15 ℃, 干燥时间为13.73 h。 结论 建立的响应面模型准确、可预测, 建立的特征图谱方法和优选的熟地黄炮制工艺方法稳定可行, 结合炮制过程的颜色变化规律, 可为熟地黄的质量控制和综合开发利用提供参考。 Abstract:OBJECTIVE To optimize the processing technique of Shudihuang (Rehmanniae Radix Praeparata, RRP) by Box-Behnken response surface model based on the change rule of characteristic spectrum and color. METHODS Multivariate statistical methods including Pearson correlation analysis, cluster analysis, principal component analysis (PCA) and orthogonal partial least squares-discriminate analysis (OPLS-DA) were used to analyze the color changes and the peak area changes of the characteristic spectrum in the processing of RRP, and to screen key quality markers (Q-markers) for RRP. Q-markers were used to evaluate the effects of steaming time, drying temperature, and drying time on the quality of RRP, and the Box-Behnken response surface method was used to determine the optimal processing technique. RESULTS The established characteristic spectrum of Shengdihuang (Rehmanniae Radix, RR) and RRP marked 18 and 20 common peaks, respectively. Among them, peak 19(5-HMF)was a new chemical component produced after the processing of RR. There was a significant difference in color (ΔE* > 12) after 2 h of evaporation, and the peak area of each characteristic was significantly different from RR. The ΔE* of RRP at different steaming time points was 0.28~4.76, indicating that the color difference could not be recognized by the naked eye. RRP from different processing points could be clustered into 3 groups with chromaticity values L*, a*, b*, E* as variables, and RRP from different processing points could be clustered into 4 groups with chromaticity values L*, a*, b*, E*, and iridoid glycosides, phenylethanolglycosides and 5-HMF as variables. The optimum processing technique of steamed RRP was as follows: moistening water was 0.3 times the amount of medicinal material, moistening time was 24 h, steaming time was 2.17 h, drying temperature was 61.15 ℃, and drying time was 13.73 h. CONCLUSION The established response surface model is accurate and predictable, the established characteristic spectrum and the optimized processing method of RRP are stable and feasible. Combining with the color change rule of the processing process, it can provide a reference for comprehensive utilization and the quality control of RRP. -
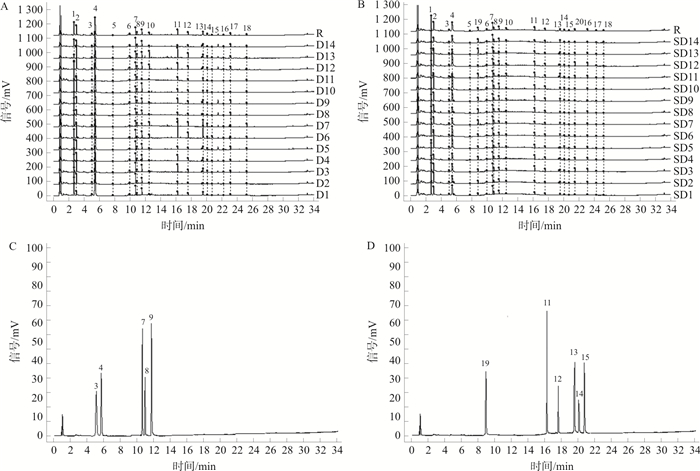
图 1 14批生地黄(A)、熟地黄(B)的叠加特征图谱及其对照特征图谱(R)、混合对照品溶液的UPLC图(C~D)
注: 3.腺苷; 4.梓醇; 7.地黄苷D; 8.地黄苷A; 9.益母草苷; 11.洋地黄叶苷C; 12.焦地黄苯乙醇苷A1;13.毛蕊花糖苷; 14.焦地黄苯乙醇苷B1;15.异毛蕊花糖苷; 19.5-HMF
Figure 1. UPLC superimposed characteristic spectrum and control characteristic spectrum (R) of 14 batches of RR (A) and RRP (B), chromatograms of mixed reference solution (C-D)
表 1 炮制过程熟地黄色度值与色差值
Table 1. Chromaticity value and color difference value of RRP during processing
编号 蒸制时间/h L* a* b* E* ΔL* Δa* Δb* ΔE* PZ1 0 17.37 8.78 14.06 24.01 / / / / PZ2 2 16.14 1.76 -2.54 16.43 -1.23 -7.02 -16.60 18.06 PZ3 4 15.69 0.57 0.29 15.70 -0.45 -1.20 2.83 3.11 PZ4 6 12.50 2.13 -2.88 13.00 -3.19 1.56 -3.18 4.76 PZ5 8 13.19 1.47 -3.57 13.75 0.69 -0.67 -0.69 1.18 PZ6 12 12.92 1.98 -3.68 13.58 -0.27 0.52 -0.12 0.60 PZ7 18 14.17 1.95 -3.39 14.70 1.24 -0.03 0.29 1.28 PZ8 24 14.12 2.18 -3.25 14.65 -0.04 0.23 0.14 0.28 PZ9 30 13.21 2.06 -3.20 13.74 -0.92 -0.12 0.05 0.93 PZ10 36 13.57 2.00 -2.91 14.03 0.37 -0.06 0.29 0.47 PZ11 42 14.30 2.10 -2.58 14.68 0.73 0.10 0.34 0.81 PZ12 48 13.06 2.75 -2.80 13.64 -1.24 0.65 -0.23 1.42 表 2 炮制过程熟地黄色度值与特征图谱各特征峰峰面积Pearson相关系数
Table 2. Pearson correlation coefficient between the chromaticity value of RRP and the peak area of each characteristic spectrum in the processing process
峰号 L* a* b* E* 1 0.546 0.316 0.360 0.490 2 0.324 0.604* 0.659* 0.564 3 0.135 -0.344 -0.175 -0.094 4 0.678* 0.632* 0.821** 0.791** 5 0.786** 0.827** 0.924** 0.938** 6 -0.413 -0.162 -0.355 -0.390 7 0.609* 0.580* 0.745** 0.715** 8 0.646* 0.633* 0.749** 0.753** 9 0.207 0.182 0.163 0.249 10 0.725** 0.872** 0.960** 0.921** 11 0.008 -0.329 -0.154 -0.143 12 0.465 -0.075 0.321 0.287 13 0.297 -0.143 0.213 0.161 14 0.496 0.097 0.435 0.409 15 0.723** 0.539 0.757** 0.755** 16 -0.307 -0.453 -0.399 -0.390 17 -0.375 -0.845** -0.620* -0.658* 18 0.174 -0.226 0.123 0.031 19 -0.123 -0.453 -0.216 -0.281 20 0.459 0.051 0.361 0.351 4+7+8+9 0.685* 0.652* 0.831** 0.804** 11+12+13+14+15 0.425 -0.026 0.329 0.298 注: *P < 0.05, * *P < 0.01。其中特征图谱4+7+8+9为环烯醚萜苷类成分总峰面积, 11+12+13+14+15为苯乙醇苷类成分总峰面积。 表 3 响应曲面试验方案和结果
Table 3. Response surface test scheme and results
序号 A/h B/℃ C/h 环烯醚萜苷类成分总峰面积 苯乙醇苷类成分总峰面积 5-HMF峰面积 Z 1 4 80 12 584 925 217 776 43 515 0.64 2 4 60 20 762 637 282 999 52 970 0.83 3 2 70 12 870 639 278 986 33 510 0.89 4 4 70 16 683 565 268 804 56 931 0.77 5 2 80 16 640 467 217 733 61 293 0.69 6 4 70 16 714 345 245 392 55 595 0.76 7 4 70 16 651 813 249 422 54 340 0.72 8 2 60 16 867 040 306 427 31 742 0.91 9 4 70 16 676 858 276 198 55 434 0.77 10 6 60 16 680 852 257 660 124 773 0.79 11 6 80 16 377 162 131 480 166 302 0.49 12 4 60 12 674 540 308 428 49 173 0.79 13 6 70 12 394 699 223 488 135 284 0.57 14 4 80 20 381 728 197 189 98 335 0.51 15 4 70 16 671 958 256 996 58 586 0.75 16 2 70 20 627 556 245 800 40 741 0.70 17 6 70 20 597 904 194 399 140 905 0.69 表 4 回归模型方差分析结果
Table 4. Regression model variance analysis results
来源 平方和 自由度 离差平方和 F值 P值 来源 平方和 自由度 离差平方和 F值 P值 模型 0.22 9 0.024 81.81 < 0.000 1 A2 2.063×10-4 1 2.063×10-4 0.69 0.433 8 A 0.053 1 0.053 176.46 < 0.000 1 B2 3.069×10-3 1 3.069×10-3 10.26 0.015 0 B 0.12 1 0.12 409.35 < 0.000 1 C2 5.012×10-3 1 5.012×10-3 16.75 0.004 6 C 3.200×10-3 1 3.200×10-3 10.69 0.013 7 残差 2.095×10-3 7 2.993×10-4 AB 1.600×10-3 1 1.600×10-3 5.35 0.054 0 失拟项 3.750×10-4 3 1.250×10-4 0.29 0.831 0 AC 0.024 1 0.024 80.27 < 0.000 1 纯误差 1.720×10-3 4 4.300×10-4 BC 7.225×10-3 1 7.225×10-3 24.14 0.001 7 总和 0.22 16 -
[1] 国家药典委员会. 中华人民共和国药典: 一部[M]. 北京: 中国医药科技出版社, 2020: 129-130.National Pharmacopoeia Committee. People's Republic of China Pharmacopoeia: Ⅰ[M]. Beijing: China medical science press, 2020: 129-130. [2] 王勃, 吕辰子, 何美菁, 等. 熟地黄炮制的研究进展[J]. 世界科学技术-中医药现代化, 2018, 20(6): 1010-1017. https://www.cnki.com.cn/Article/CJFDTOTAL-SJKX201806031.htmWANG B, LYU CZ, HE MJ, et al. Research progress on rehmanniae Radix preparata[J]. Mod Tradit Chin Med Mater Med World Sci Technol, 2018, 20(6): 1010-1017. https://www.cnki.com.cn/Article/CJFDTOTAL-SJKX201806031.htm [3] 葛楠, 闫广利, 孙晖, 等. 熟地黄药效物质基础研究进展[J]. 中草药, 2023, 54(1): 292-302. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202301031.htmGE N, YAN GL, SUN H, et al. Research progress on effective constituents in Radix Rehmanniae Praeparata[J]. Chin Tradit Herb Drugs, 2023, 54(1): 292-302. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202301031.htm [4] 朱珏, 朱香梅, 石雨荷, 等. 地黄的研究进展及其质量标志物的预测分析[J]. 中药材, 2022, 45(5): 1273-1281. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYHS202307032.htmZHU J, ZHU XM, SHI YH, et al. Research progress of Rehmannia glutinosa and prediction and analysis of its quality markers[J]. J Chin Med Mater, 2022, 45(5): 1273-1281. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYHS202307032.htm [5] 张留记, 王建霞, 屠万倩, 等. 生地黄与熟地黄中5个苷类成分和总多糖的含量比较[J]. 天然产物研究与开发, 2019, 31(4): 566-571. https://www.cnki.com.cn/Article/CJFDTOTAL-TRCW201904002.htmZHANG LJ, WANG JX, TU WQ, et al. Comparation of the contents of 5 glycosides and total polysaccharides in Rehmanniae Radix and Rehmanniae Radix Praeparata[J]. Nat Prod Res Dev, 2019, 31(4): 566-571. https://www.cnki.com.cn/Article/CJFDTOTAL-TRCW201904002.htm [6] 沈丽琴, 杨晗, 李胜华, 等. 地黄不同工艺炮制过程中主要活性成分的变化规律研究[J]. 亚太传统医药, 2019, 15(10): 75-79. https://www.cnki.com.cn/Article/CJFDTOTAL-YTCT201910023.htmSHEN LQ, YANG H, LI SH, et al. Study on variation regularity of main active components in Radix rehmanniae steamed with different processing methods[J]. Asia Pac Tradit Med, 2019, 15(10): 75-79. https://www.cnki.com.cn/Article/CJFDTOTAL-YTCT201910023.htm [7] 李娴, 邢亚东, 李姗姗, 等. 不同炮制方法对熟地黄中7种化学成分含量的影响[J]. 蚌埠医学院学报, 2020, 45(5): 634-637, 643. https://www.cnki.com.cn/Article/CJFDTOTAL-BANG202005022.htmLI X, XING YD, LI SS, et al. Effect of different processing methods on the contents of seven chemical components in Rehmanniae Radix Praeparata[J]. J Bengbu Med Coll, 2020, 45(5): 634-637, 643. https://www.cnki.com.cn/Article/CJFDTOTAL-BANG202005022.htm [8] 刘彦飞, 赵宇, 武卫红, 等. 地黄的化学成分及其在加工炮制过程中的变化[J]. 国外医药(植物药分册), 2007, 22(3): 102-108. https://www.cnki.com.cn/Article/CJFDTOTAL-GWZW200703003.htmLIU YF, ZHAO Y, WU WH, et al. Chemical constituents of Rehmannia glutinosa and their changes during processing[J]. World Phytomedicines, 2007, 22(3): 102-108. https://www.cnki.com.cn/Article/CJFDTOTAL-GWZW200703003.htm [9] 张文婷, 岳超, 黄琴伟, 等. 地黄生品与炮制品中8个糖类成分及不同炮制时间点其量变化分析[J]. 中草药, 2016, 47(7): 1132-1136. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201607017.htmZHANG WT, YUE C, HUANG QW, et al. Contents of eight saccharides in unprocessed and processed Rehmannia glutinosa and content changes at different processing time points[J]. Chin Tradit Herb Drugs, 2016, 47(7): 1132-1136. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201607017.htm [10] 吕思霖. 基于性状、化学成分的地黄九蒸九晒炮制科学性研究[D]. 太原: 山西大学, 2021.LYU SL. Scientific research on the processing of Rehmannia glutinosa by nine steaming and nine Sun drying based on characters and chemical components[D]. Taiyuan: Shanxi University, 2021. [11] 解杨, 钟凌云, 薛晓, 等. 基于多指标-响应曲面法优选炆地黄炮制工艺及成分与色泽相关性分析[J]. 中国中药杂志, 2022, 47(18): 4927-4937. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202218011.htmXIE Y, ZHONG LY, XUE X, et al. Optimization of processing technology of braised Rehmanniae Radix based on multiple indexes and response surface technology and correlation between components and color[J]. China J Chin Mater Med, 2022, 47(18): 4927-4937. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202218011.htm [12] 国家药典委员会. 中华人民共和国药典: 四部[M]. 北京: 中国医药科技出版社, 2020: 32.National Pharmacopoeia Committee. People's Republic of China Pharmacopoeia: Ⅳ[M]. Beijing: China medical science press, 2020: 32. [13] 钟文峰, 洪婉敏, 刘晓霞, 等. 基于特征图谱结合效毒值与外观性状的蜜马兜铃炮制工艺研究[J]. 南京中医药大学学报, 2023, 39(2): 157-169. doi: 10.14148/j.issn.1672-0482.2023.0157ZHONG WF, HONG WM, LIU XX, et al. Study on processing technology of honey Aristolochia based on characteristic map combined with toxicity value and appearance characteristics[J]. J Nanjing Univ Tradit Chin Med, 2023, 39(2): 157-169. doi: 10.14148/j.issn.1672-0482.2023.0157 [14] 严斐霞, 谢永艳, 陈畅, 等. 熟地黄炮制过程中的化学成分变化和药理作用研究进展[J]. 时珍国医国药, 2021, 32(10): 2493-2495. https://www.cnki.com.cn/Article/CJFDTOTAL-SZGY202110050.htmYAN FX, XIE YY, CHEN C, et al. Research progress on chemical composition changes and pharmacological effects of Radix Rehmanniae Preparata during processing[J]. Lishizhen Med Mater Med Res, 2021, 32(10): 2493-2495. https://www.cnki.com.cn/Article/CJFDTOTAL-SZGY202110050.htm [15] 王梅, 王越欣, 武英茹, 等. 5-羟甲基糠醛在中药加工过程中的变化及药理作用研究概况[J]. 药物评价研究, 2020, 43(11): 2319-2327. https://www.cnki.com.cn/Article/CJFDTOTAL-YWPJ202011030.htmWANG M, WANG YX, WU YR, et al. Research progress on content changes of 5-hydroxymethyl furfural during processing of Chinese medicine and its pharmacological effects[J]. Drug Eval Res, 2020, 43(11): 2319-2327. https://www.cnki.com.cn/Article/CJFDTOTAL-YWPJ202011030.htm [16] 于文娜, 张振凌, 张颖, 等. 地黄炮制过程中异毛蕊花糖苷含量的动态变化[J]. 中国实验方剂学杂志, 2017, 23(18): 22-26. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201718005.htmYU WN, ZHANG ZL, ZHANG Y, et al. Determination of dynamic changes of isoacteoside in processing process of rehmanniae Radix[J]. Chin J Exp Tradit Med Formulae, 2017, 23(18): 22-26. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201718005.htm [17] 陈金鹏, 张克霞, 刘毅, 等. 地黄化学成分和药理作用的研究进展[J]. 中草药, 2021, 52(6): 1772-1784. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202106029.htmCHEN JP, ZHANG KX, LIU Y, et al. Research progress on chemical constituents and pharmacological actions of Rehmannia glutinosa[J]. Chin Tradit Herb Drugs, 2021, 52(6): 1772-1784. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202106029.htm [18] 国家药典委员会. 中华人民共和国药典: 一部[M]. 北京: 中国医药科技出版社, 2015: 124-126.National Pharmacopoeia Committee. People's Republic of China Pharmacopoeia: Ⅰ[M]. Beijing: China medical science press, 2015: 124-126. -





 下载:
下载: