Mechanism of Yi Ceng Application in Alleviating Synovial Fibrosis Induced by Transforming Growth Factor-β1 in Rats with Knee Osteoarthritis Based on PI3K/AKT/HIF-1α Signal Pathway
-
摘要:
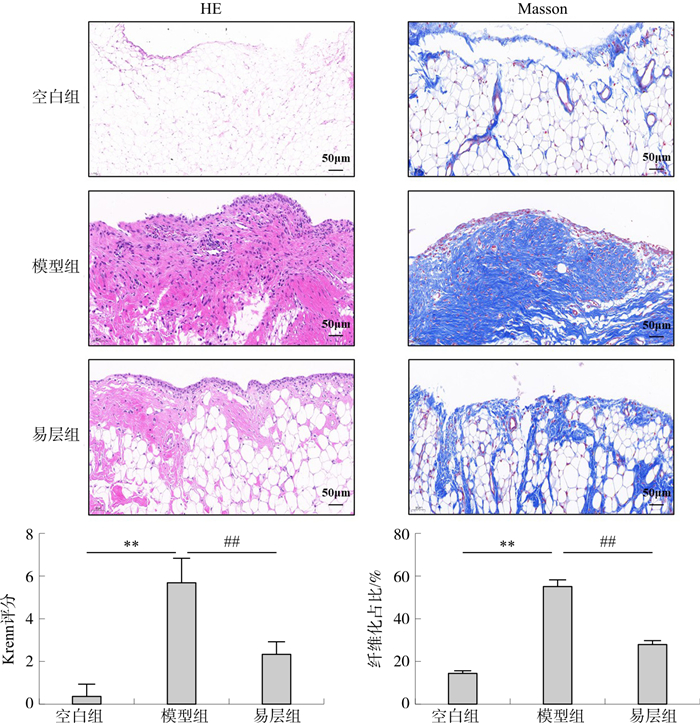
目的 探讨易层敷贴对膝骨关节炎(KOA)大鼠滑膜纤维化TGF-β1、α-SMA、COL1A1表达的影响以及对PI3K/AKT/HIF-1α信号通路的调控作用。 方法 将30只SPF级SD大鼠随机分为空白组、模型组和易层组, 膝关节腔注射200 ng转化生长因子-β1重组蛋白(TGF-β1)建立膝关节滑膜纤维化动物模型, 2 d 1次, 持续3次, 造模14 d后给予易层敷贴外用治疗28 d后取滑膜组织。HE和Masson染色观察滑膜病理变化; 免疫组化检测滑膜p-PI3K、p-AKT、HIF-1α、TGF-β1、α-SMA、COL1A1的表达水平。提取雄性SD大鼠膝关节成纤维样滑膜细胞(FLSs), 以10 ng·mL-1 TGF-β1诱导24 h建立KOA滑膜纤维化细胞模型, 易层冻干粉干预24 h, Western blot检测PI3K、p-PI3K、AKT、p-AKT、HIF-1α、TGF-β1、α-SMA、COL1A1的蛋白表达, qPCR检测HIF-1α、TGF-β1、α-SMA、COL1A1 mRNA表达。 结果 与空白组相比, 模型组HE染色滑膜炎加重(P < 0.01), Masson染色滑膜纤维化占比显著增加(P < 0.01), 免疫组化中p-PI3K、p-AKT、HIF-1α、TGF-β1、α-SMA、COL1A1表达均增加(P < 0.01);滑膜细胞中p-PI3K/PI3K、p-AKT/AKT、HIF-1α、TGF-β1、α-SMA、COL1A1蛋白表达均升高(P < 0.05, P < 0.01), HIF-1α、TGF-β1、α-SMA、COL1A1 mRNA表达均上升(P < 0.01)。与模型组相比, 易层组滑膜炎和滑膜纤维化状况改善, p-PI3K、p-AKT、HIF-1α、TGF-β1、α-SMA、COL1A1的表达均减少(P < 0.05, P < 0.01);滑膜细胞中p-PI3K/PI3K、p-AKT/AKT、HIF-1α、TGF-β1、α-SMA、COL1A1蛋白表达均降低(P < 0.05), HIF-1α、TGF-β1、α-SMA、COL1A1 mRNA表达均下调(P < 0.05, P < 0.01)。 结论 易层敷贴通过调控PI3K/AKT/HIF-1α信号通路, 降低TGF-β1、α-SMA、COL1A1的表达, 有效改善TGF-β1诱导的KOA大鼠滑膜纤维化。 -
关键词:
- 膝骨关节炎 /
- 滑膜纤维化 /
- PI3K/AKT/HIF-1α信号通路 /
- 易层敷贴
Abstract:OBJECTIVE To investigate the effect of Yi Ceng application on the expression of TGF-β1, α-SMA and COL1A1 in synovial fibrosis of rats with knee osteoarthritis (KOA) and the regulation of PI3K/AKT/HIF-1α signal pathway. METHODS 30 SPF SD rats were randomly divided into blank group, model group and Yi Ceng group. The animal model of synovial fibrosis of knee joint was established with 200 ng transforming growth factor-β1 (TGF-β1) recombinant protein, once every 2 days, lasting for 3 times. After 14 days of modeling, the synovial tissue was obtained after the external application of Yi Ceng for 28 days. HE and Masson staining were used to observe the pathological changes of synovium. The expression of p-PI3K, p-AKT, HIF-1α, TGF-β1, α-SMA and COL1A1 in synovium was detected by immunohistochemistry. Fibroblast-like synoviocytes (FLSs) were isolated from the knee joint of male SD rats and induced with 10 ng·mL-1 TGF-β1 for 24 h to establish KOA synovial fibrosis cell model, then intervened with Yi Ceng freeze-dried powder for 24 h. PI3K, p-PI3K, AKT, p-AKT, HIF-1α, TGF-β1, α-SMA and COL1A1 protein expressions in synoviocytes were detected by Western blot. HIF-1α, TGF-β1, α-SMA and COL1A1 mRNA expressions in synoviocytes were detected by qPCR. RESULTS Compared with the control group, the synovitis and synovial fibrosis of the model group were significantly aggravated (P < 0.01) in HE and Masson staining, and the expressions of p-PI3K, p-AKT, HIF-1α, TGF-β1, α-SMA and COL1A1 were increased (P < 0.01). The protein expressions of p-PI3K/PI3K, p-AKT/AKT, HIF-1α, TGF-β1, α-SMA and COL1α1 in synoviocytes were increased (P < 0.05, P < 0.01), and the mRNA expressions of HIF-1α, TGF-β1, α-SMA and COL1A1 in synoviocytes were increased (P < 0.01). Compared with the model group, synovitis and synovial fibrosis were improved, and the expression levels of p-PI3K, p-AKT, HIF-1α, TGF-β1, α-SMA and COL1A1 were decreased in the Yi Ceng group (P < 0.05, P < 0.01).The protein expressions of p-PI3K/PI3K, p-AKT/AKT, HIF-1α, TGF-β1, α-SMA and COL1A1 were decreased (P < 0.05), and mRNA levels of HIF-1α, TGF-β1, α-SMA and COL1A1 were downregulated (P < 0.05, P < 0.01). CONCLUSION Yi Ceng application can effectively improve the synovial fibrosis induced by recombinant TGF-β1 in KOA rats by decreasing the expression of TGF-β1, α-SMA and COL1A1 via the PI3K/AKT/HIF-1α signal pathway. -
表 1 目的基因序列
Table 1. Sequence of target genes
基因 序列(5′→3′) HIF-1α Forward
ReverseAACCCATCAACTCAGTAATC
CTACAAGAAACCGCCTATTGF-β1 Forward
ReverseCTCAGGCGTATCAGTGGG
ATGGTGGACCGCAACAACα-SMA Forward
ReverseGCTGTTATAGGTGGTTTCG
ACTGCTGCTTCCTCTTCTTCOL1A1 Forward
ReverseCCAGGGAGACCGTTGAGT
ACAAGGTGACAGAGGCATAAAβ-actin Forward
ReverseGAGAGGGAAATCGTGCGT
GGAGGAAGAGGATGCGG -
[1] LI DH, LI SJ, CHEN Q, et al. The prevalence of symptomatic knee osteoarthritis in relation to age, sex, area, region, and body mass index in China: A systematic review and meta-analysis[J]. Front Med, 2020, 7: 304. doi: 10.3389/fmed.2020.00304 [2] MATHIESSEN A, CONAGHAN PG. Synovitis in osteoarthritis: Current understanding with therapeutic implications[J]. Arthritis Res Ther, 2017, 19(1): 18. doi: 10.1186/s13075-017-1229-9 [3] ZHANG L, LI MC, LI XC, et al. Characteristics of sensory innervation in synovium of rats within different knee osteoarthritis models and the correlation between synovial fibrosis and hyperalgesia[J]. J Adv Res, 2021, 35: 141-151. [4] WEI QL, KONG N, LIU XH, et al. Pirfenidone attenuates synovial fibrosis and postpones the progression of osteoarthritis by anti-fibrotic and anti-inflammatory properties in vivo and in vitro[J]. J Transl Med, 2021, 19(1): 157. doi: 10.1186/s12967-021-02823-4 [5] CHEN GY, LIU XY, CHEN JQ, et al. Prediction of rhizoma drynariae targets in the treatment of osteoarthritis based on network pharmacology and experimental verification[J]. Evid Based Complement Alternat Med, 2021, 2021: 5233462. [6] MA YC, BAO YN, WU LF, et al. IL-8 exacerbates CCl4-induced liver fibrosis in human IL-8-expressing mice via the PI3K/Akt/HIF-1α pathway[J]. Mol Immunol, 2022, 152: 111-122. doi: 10.1016/j.molimm.2022.10.011 [7] ZHOU SS, AI ZZ, LI WN, et al. Deciphering the pharmacological mechanisms of taohe-chengqi decoction extract against renal fibrosis through integrating network pharmacology and experimental validation in vitro and in vivo[J]. Front Pharmacol, 2020, 11: 425. doi: 10.3389/fphar.2020.00425 [8] 吴鹏, 黄正泉, 邢润麟, 等. 基于HMGB1探讨"易层"贴敷对KOA大鼠滑膜纤维化的影响[J]. 南京中医药大学学报, 2019, 35(5): 547-551. http://xb.njucm.edu.cn/article/id/zr20190513WU P, HUANG ZQ, XING RL, et al. Effect of yiceng on the synovial fibrosis in KOA rats based on HMGB1[J]. J Nanjing Univ Tradit Chin Med, 2019, 35(5): 547-551. http://xb.njucm.edu.cn/article/id/zr20190513 [9] VAN BEUNINGEN HM, GLANSBEEK HL, VAN DER KRAAN PM, et al. Osteoarthritis-like changes in the murine knee joint resulting from intra-articular transforming growth factor-beta injections[J]. Osteoarthritis Cartil, 2000, 8(1): 25-33. doi: 10.1053/joca.1999.0267 [10] KRENN V, MORAWIETZ L, BURMESTER GR, et al. Synovitis score: Discrimination between chronic low-grade and high-grade synovitis[J]. Histopathology, 2006, 49(4): 358-364. doi: 10.1111/j.1365-2559.2006.02508.x [11] LIAO B, GUAN MT, TAN QY, et al. Low-intensity pulsed ultrasound inhibits fibroblast-like synoviocyte proliferation and reduces synovial fibrosis by regulating Wnt/β-catenin signaling[J]. J Orthop Translat, 2021, 30: 41-50. doi: 10.1016/j.jot.2021.08.002 [12] 丁春明. 基于古今文献积聚研究及"纤维化"中医病机理论探讨[D]. 哈尔滨: 黑龙江中医药大学, 2018.DING CM. Based on the research of ancient and modern literature accumulation and the discussion of TCM pathogenesis theory of "fibrosis"[D]. Harbin: Heilongjiang University of Chinese Medicine, 2018. [13] 向璐, 张巧艳, 赵琦明, 等. 黄芪-当归化学成分、药理作用及临床应用的研究进展[J]. 中草药, 2022, 53(7): 2196-2213. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202207030.htmXIANG L, ZHANG QY, ZHAO QM, et al. Research progress on chemical constituents, pharmacological effects and clinical applications of Astragali Radix-Angelicae Sinensis Radix[J]. Chin Tradit Herb Drugs, 2022, 53(7): 2196-2213. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202207030.htm [14] 宋丹君, 吕晓东, 庞立健, 等. 基于数据挖掘与网络药理学探究中医药治疗肺纤维化用药规律及作用机制[J]. 辽宁中医药大学学报, 2023, 25(6): 44-51. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202220026.htmSONG DJ, LYU XD, PANG LJ, et al. Analysis of medication rules and mechanisms of Chinese medicine in treatment of pulmonary fibrosis based on data mining and network pharmacology[J]. J Liaoning Univ Tradit Chin Med, 2023, 25(6): 44-51. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202220026.htm [15] 王培民, 黄国淳, 丁亮, 等. "易层"贴敷疗法治疗膝骨性关节炎的临床疗效观察[J]. 世界科学技术-中医药现代化, 2014, 16(10): 2097-2101. doi: 10.11842/wst.2014.10.007WANG PM, HUANG GC, DING L, et al. Clinic effect of layers adjusting external application therapy on knee osteoarthritis[J]. Mod Tradit Chin Med Mater Med World Sci Technol, 2014, 16(10): 2097-2101. doi: 10.11842/wst.2014.10.007 [16] REMST DFG, BLANEY DAVIDSON EN, VAN DER KRAAN PM. Unravelling osteoarthritis-related synovial fibrosis: A step closer to solving joint stiffness[J]. Rheumatology, 2015, 54(11): 1954-1963. doi: 10.1093/rheumatology/kev228 [17] ZHANG TZ, HE XL, CALDWELL L, et al. NUAK1 promotes organ fibrosis via YAP and TGF-β/SMAD signaling[J]. Sci Transl Med, 2022, 14(637): eaaz4028. [18] XIANG DJ, ZOU J, ZHU XY, et al. Physalin D attenuates hepatic stellate cell activation and liver fibrosis by blocking TGF-β/Smad and YAP signaling[J]. Phytomedicine, 2020, 78: 153294. doi: 10.1016/j.phymed.2020.153294 [19] HU HH, CHEN DQ, WANG YN, et al. New insights into TGF-β/Smad signaling in tissue fibrosis[J]. Chem Biol Interact, 2018, 292: 76-83. doi: 10.1016/j.cbi.2018.07.008 [20] KONG P, SHINDE AV, SU Y, et al. Opposing actions of fibroblast and cardiomyocyte Smad3 signaling in the infarcted myocardium[J]. Circulation, 2018, 137(7): 707-724. doi: 10.1161/CIRCULATIONAHA.117.029622 [21] PENG DD, FU MY, WANG MN, et al. Targeting TGF-β signal transduction for fibrosis and cancer therapy[J]. Mol Cancer, 2022, 21(1): 104. doi: 10.1186/s12943-022-01569-x [22] WU LW, ZHANG QH, MO WH, et al. Quercetin prevents hepatic fibrosis by inhibiting hepatic stellate cell activation and reducing autophagy via the TGF-β1/Smads and PI3K/Akt pathways[J]. Sci Rep, 2017, 7(1): 9289. doi: 10.1038/s41598-017-09673-5 [23] 陈炜, 袁平凡, 张念志. 益气养阴活血法对博来霉素诱导IPF的大鼠PI3K/AKT/HIF-1α信号通路的影响[J]. 时珍国医国药, 2021, 32(7): 1577-1581. https://www.cnki.com.cn/Article/CJFDTOTAL-SZGY202107010.htmCHEN W, YUAN PF, ZHANG NZ. Effect of Yiqi Yangyin Huoxue Method on PI3K/AKT/HIF-1α signaling pathway in rats with bleomycin-induced IPF[J]. Lishizhen Med Mater Med Res, 2021, 32(7): 1577-1581. https://www.cnki.com.cn/Article/CJFDTOTAL-SZGY202107010.htm [24] YANG R, TANG YY, HOU JY, et al. Fibrosis in frozen shoulder: Activation of IL-6 through PI3K-Akt signaling pathway in synovial fibroblast[J]. Mol Immunol, 2022, 150: 29-38. [25] ZHANG L, ZHANG L, HUANG ZQ, et al. Increased HIF-1 α in knee osteoarthritis aggravate synovial fibrosis via fibroblast-like synoviocyte pyroptosis[J]. Oxid Med Cell Longev, 2019, 2019: 6326517. -





 下载:
下载: