Ethanol Extract of Tengligen Induces Apoptosis of Colorectal Cancer Cells through AKT/PUMA Pathway
-
摘要:
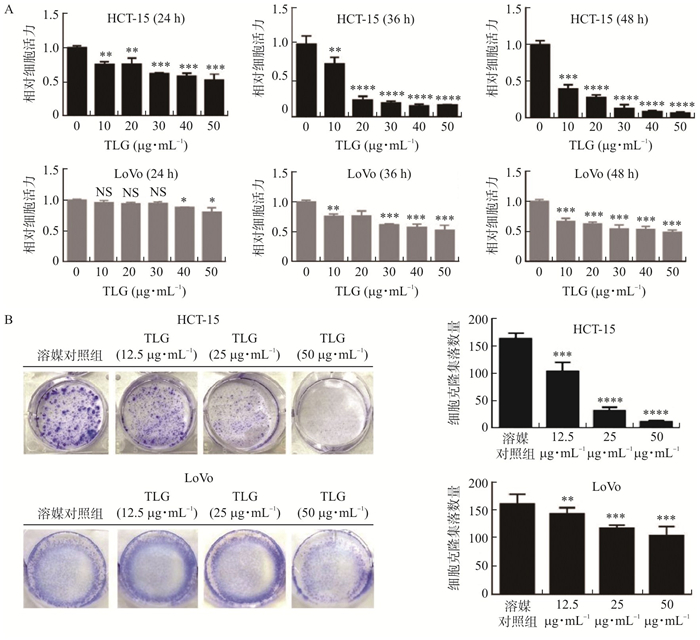
目的 研究藤梨根乙醇提取物(TLG)对结直肠癌细胞的作用及其抗肿瘤机制。 方法 利用MTT检测TLG对人结直肠腺癌细胞HCT-15、LoVo的毒性, 细胞平板克隆实验检测TLG对结直肠癌细胞HCT-15、LoVo增殖的影响, Hoechst-33258染色法和流式细胞术检测TLG对结直肠癌细胞凋亡的影响, Western blot实验检测TLG对HCT-15中相关蛋白Caspase-3、PARP、P65、PUMA、AKT、GSK-3β表达的影响, 双荧光素酶报告基因实验检测TLG对HCT-15中相关启动子活性的影响。 结果 结直肠癌细胞HCT-15、LoVo经TLG处理后, 与对照组相比, 细胞活力以时间及剂量依赖性下降(P < 0.01,P < 0.001), 转染AKT cDNA、PUMA siRNA或GSK-3β抑制剂处理后能够恢复增殖(P < 0.001);不同浓度TLG处理后, 细胞凋亡率提高(P < 0.01, P < 0.001), 转染AKT cDNA或GSK-3β抑制剂处理后能够遏制TLG诱导的凋亡(P < 0.01,P < 0.001);Western blot显示HCT-15细胞中的p-AKT/AKT、p-GSK-3β/GSK-3β蛋白表达量随TLG浓度上升而减少(P < 0.01), Cleaved-Caspase-3/Caspase-3、Cleaved-PARP/PARP、p65、PUMA蛋白表达量随TLG浓度上升而增加(P < 0.05, P < 0.01,P < 0.001), 同时转染AKT cDNA、PUMA siRNA或GSK-3β抑制剂处理后能够阻滞TLG对下游蛋白Cleaved-Caspase-3/Caspase-3、Cleaved-PARP/PARP、p65、PUMA的影响(P < 0.05, P < 0.01,P < 0.001);报告基因结果显示NF-κB、PUMA启动子活性随TLG浓度上升而增强(P < 0.05, P < 0.01), 同时转染AKT cDNA或GSK-3β抑制剂处理后恢复(P < 0.01, P < 0.001)。 结论 TLG可以显著诱导结直肠癌细胞凋亡, 发挥较强的抑制肿瘤作用, 其机制可能与AKT/PUMA信号通路有关。 Abstract:OBJECTIVE To investigate the effect and antitumor mechanism of ethanol extract of Tengligen (TLG) on colorectal cancer cells. METHODS The toxicity of TLG on human colorectal adenocarcinoma cells (HCT-15, LoVo) was detected by methyl thiazolyl tetrazolium (MTT). Cell plate cloning assay was used to detect the effect of TLG on colorectal cancer cell proliferation. Hoechst-33258 staining and flow cytometry were used to detect the effect of TLG on colorectal cancer cell apoptosis. Western blot assay was used to detect the effect of TLG on the related proteins Caspase-3, PARP, p65, PUMA, AKT, GSK-3β in HCT-15. Dual luciferase reporter assay was performed to detect the effect of TLG on the activity of related promoter in HCT-15. RESULTS Treatment of colorectal cancer cells (HCT-15, LoVo) with TLG resulted in a time- and dose-dependent decrease (P < 0.01, P < 0.001) in cell viability compared with control cells, and transfection with AKT cDNA, PUMA siRNA or GSK-3β inhibitor treatment was able to restore proliferation (P < 0.01). After treatment with different concentrations of TLG, the rate of apoptosis was increased (P < 0.01, P < 0.001), and cells transfected with AKT cDNA, PUMA siRNA, or GSK-3β inhibitor was able to suppress TLG induced apoptosis (P < 0.01, P < 0.001). Western blot showed that p-AKT/AKT and p-GSK-3β/GSK-3β protein expression in HCT-15 cells decreased (P < 0.001) while Cleaved-Caspase-3/Caspase-3, Cleaved PARP/PARP, p65 and PUMA protein levels increased with the increase of TLG concentrations (P < 0.05, P < 0.01, P < 0.001); simultaneous transfection of AKT cDNA, PUMA siRNA, or GSK-3β inhibitor was able to block the effects of TLG on downstream proteins Cleaved-Caspase-3/Caspase-3, Cleaved PARP/PARP, p65, PUMA (P < 0.05, P < 0.01, P < 0.001). Reporter gene results showed that NF-κB and PUMA promoter activities were enhanced (P < 0.05, P < 0.01) with the increase in TLG concentration, but recovered by simultaneous transfection of AKT cDNA or GSK-3β inhibitor treatment (P < 0.01, P < 0.001). CONCLUSION TLG can significantly induce apoptosis and exert strong tumor suppressive effects in colorectal cancer cells, and the mechanism may be related to the AKT/PUMA signaling pathway. -
Key words:
- ethanol extract of Tengligen /
- colorectal cancer /
- cell apoptosis /
- AKT /
- GSK-3β /
- NF-κB /
- PUMA
-
图 4 TLG诱导结直肠癌细胞HCT-15细胞系凋亡与NF-κB/PUMA通路有关
注:A.Western blot实验检测不同浓度TLG对HCT-15细胞中p65、PUMA蛋白表达水平调控的情况; B~C.双荧光素酶报告基因实验检测不同浓度TLG对HCT-15细胞中NF-κB、PUMA启动子的影响; D.Western blot实验检测HCT-15细胞在利用PUMA siRNA敲低PUMA蛋白表达后, 对下游Cleaved-Caspase-3/Caspase-3、Cleaved-PARP/PARP的表达影响; E.MTT实验检测HCT-15细胞在利用PUMA siRNA敲低PUMA蛋白表达后, TLG对HCT-15细胞的抑制效果改变情况。与溶媒对照组比较, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.000 1;与50 μg·mL-1 TLG组比较,###P < 0.001。x±s,n=3。
Figure 4. TLG induced apoptosis in human colorectal cancer cell line HCT-15 by NF-κB/PUMA pathway
图 5 TLG可能通过AKT/GSK-3β信号通路影响NF-κB/PUMA通路
注:A.Western blot实验检测不同浓度TLG对HCT-15细胞中p-AKT/AKT、p-GSK-3β/GSK-3β蛋白表达水平调控的情况; B~C.双荧光素酶报告基因实验检测HCT-15细胞过表达AKT蛋白(GSK-3β抑制剂)同时TLG加药处理对NF-κB、PUMA启动子的影响; D~E.Western blot实验检测HCT-15细胞过表达AKT蛋白(GSK-3β抑制剂)同时TLG加药处理对下游蛋白的表达影响; 与溶媒对照组比较, **P < 0.01, ***P < 0.001;与50 μg·mL-1 TLG组比较,##P < 0.01,###P < 0.001。x±s,n=3。
Figure 5. TLG may affect NF-κB/PUMA pathway by AKT/GSK-3β signaling pathway
图 6 GSK-3β抑制剂和AKT-cDNA能够有效遏制TLG对HCT-15的杀伤效果
注: A~B.MTT实验检测HCT-15细胞在利用AKT cDNA过表达及GSK-3β抑制剂处理后, TLG对HCT-15细胞活力的抑制效果改变情况; C.平板克隆实验检测HCT-15细胞在利用AKT cDNA过表达及GSK-3β抑制剂处理后, TLG对HCT-15细胞克隆集落数量的抑制效果改变情况; D.Hoechst-33258染色实验检测HCT-15细胞在利用AKT cDNA过表达及GSK-3β抑制剂处理后, TLG对HCT-15细胞凋亡的诱导效果改变情况; 与溶媒对照组比较, **P < 0.01, ***P < 0.001;与50 μg·mL-1 TLG组比较,##P < 0.01,###P < 0.001。
Figure 6. Utilization of GSK-3β inhibitor and overexpression of AKT cDNA to effectively suppress the killing effect of TLG on HCT-15
-
[1] SUNG H, FERLAY J, SIEGEL RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660 [2] ARNOLD M, SIERRA MS, LAVERSANNE M, et al. Global patterns and trends in colorectal cancer incidence and mortality[J]. Gut, 2017, 66(4): 683-691. doi: 10.1136/gutjnl-2015-310912 [3] MASSIMO LOPEZ MD. Adjuvant therapy of colorectal cancer[J]. Dis Colon Rectum, 1994, 37(2): S86-S91. doi: 10.1007/BF02048438 [4] EILSTEIN D, HEDELIN G, SCHAFFER P. Incidence of colorectal cancer in Bas-Rhin, trend and prediction in 2009[J]. Bull Cancer, 2000, 87(7/8): 595-599. [5] 张新, 孙华, 李亚东. 孙桂芝治疗大肠癌经验[J]. 山东中医杂志, 1998, 17(4): 173-175. https://www.cnki.com.cn/Article/CJFDTOTAL-SDZY804.023.htmZHANG X, SUN H, LI YD. Sun Guizhi's experience in treating colorectal cancer[J]. Shandong J Tradit Chin Med, 1998, 17(4): 173-175. https://www.cnki.com.cn/Article/CJFDTOTAL-SDZY804.023.htm [6] 李博林, 王志坤, 张纨, 等. 刘启泉治疗胃癌经验[J]. 中医杂志, 2014, 55(17): 1454-1456. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYZ201417005.htmLI BL, WANG ZK, ZHANG W, et al. LIU Qiquan's experience in treating stomach cancer[J]. J Tradit Chin Med, 2014, 55(17): 1454-1456. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYZ201417005.htm [7] 刘文佳, 齐元富. 齐元富运用补中益气汤辨治肝癌验案1则[J]. 湖南中医杂志, 2018, 34(6): 113-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZO201806063.htmLIU WJ, QI YF. Qi Yuanfu's experience in treating liver cancer by Buzhong Yiqi Decoction: A case report[J]. Hunan J Tradit Chin Med, 2018, 34(6): 113-114. https://www.cnki.com.cn/Article/CJFDTOTAL-HNZO201806063.htm [8] 马丽雅. 四君子汤加减在抗肿瘤中的应用[J]. 浙江中医学院学报, 2004, 28(2): 44. https://www.cnki.com.cn/Article/CJFDTOTAL-BHON200402027.htmMA LY. Application of modified Sijunzi Decoction in anti-tumor[J]. J Zhejiang Coll Tcm, 2004, 28(2): 44. https://www.cnki.com.cn/Article/CJFDTOTAL-BHON200402027.htm [9] 吴霜霜, 戚益铭, 阮善明, 等. 基于系统聚类分析总结吴良村辨治结直肠癌经验[J]. 中华中医药杂志, 2017, 32(1): 142-145. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY201701037.htmWU SS, QI YM, RUAN SM, et al. WU Liangcun's experience in differentiating and treating rectal carcinoma based on cluster analysis[J]. China J Tradit Chin Med Pharm, 2017, 32(1): 142-145. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY201701037.htm [10] 何国浓, 王邦才, 王辉, 等. 中药藤梨根提取物体外抗肿瘤活性部位的筛选[J]. 中华中医药杂志, 2017, 32(6): 2683-2685. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY201706096.htmHE GN, WANG BC, WANG H, et al. Screening in vitro anti-tumor active parts of Radix Actinidiae extract[J]. China J Tradit Chin Med Pharm, 2017, 32(6): 2683-2685. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY201706096.htm [11] 滕坤, 张海丰, 臧皓, 等. 藤梨根甲醇提取物的抗氧化活性及物质基础初步研究[J]. 中草药, 2019, 50(18): 4384-4388. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201918021.htmTENG K, ZHANG HF, ZANG H, et al. Antioxidant activities in vitro and components analysis of methanol extract of Actinidia arguta by HPLC-DAD-ESI-MS/MS[J]. Chin Tradit Herb Drugs, 2019, 50(18): 4384-4388. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201918021.htm [12] 欧阳红涛, 吴晶莹, 陈玉祥. 藤梨根抗病毒活性部位的初步筛选[J]. 中国现代医学杂志, 2011, 21(4): 404-407. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXDY201104008.htmOUYANG HT, WU JY, CHEN YX. Preliminary screening of Actinidiae antiviral activity fraction[J]. China J Mod Med, 2011, 21(4): 404-407. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXDY201104008.htm [13] 陈军, 华维一, 孙宏斌. 积雪草酸及其衍生物的生物活性研究概况[J]. 中草药, 2006, 37(3): 458-460. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO200603051.htmCHEN J, HUA WY, SUN HB. Advances in studies on biological activities of Asiatic acid and its derivatives[J]. Chin Tradit Herb Drugs, 2006, 37(3): 458-460. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO200603051.htm [14] 王晓戎, 袁孝兵, 李平, 等. 大肠癌中医证候分布临床调查研究[J]. 长春中医药大学学报, 2011, 27(3): 377-379. https://www.cnki.com.cn/Article/CJFDTOTAL-CZXX201103023.htmWANG XR, YUAN XB, LI P, et al. Clinical investigation and study on TCM syndrome distribution of colorectal cancer[J]. J Chang Univ Tradit Chin Med, 2011, 27(3): 377-379. https://www.cnki.com.cn/Article/CJFDTOTAL-CZXX201103023.htm [15] WANG HN, ZHANG LL, YANG X, et al. PUMA mediates the combinational therapy of 5-FU and NVP-BEZ235 in colon cancer[J]. Oncotarget, 2015, 6(16): 14385-14398. [16] SZEPESHAZI K, SCHALLY AV, HALMOS G, et al. Targeted cytotoxic somatostatin analogue AN-238 inhibits somatostatin receptor-positive experimental colon cancers independently of their p53 status[J]. Cancer Res, 2002, 62(3): 781-788. [17] DUDGEON C, PENG R, WANG P, et al. Inhibiting oncogenic signaling by sorafenib activates PUMA via GSK3β and NF-κB to suppress tumor cell growth[J]. Oncogene, 2012, 31(46): 4848-4858. [18] LI LJ, LIN L, LI M, et al. Gilteritinib induces PUMA-dependent apoptotic cell death via AKT/GSK-3β/NF-κB pathway in colorectal cancer cells[J]. J Cell Mol Med, 2020, 24(3): 2308-2318. [19] QIAO M, SHENG SJ, PARDEE AB. Metastasis and AKT activation[J]. Cell Cycle, 2008, 7(19): 2991-2996. [20] TONG YX, PARK S, WU D, et al. Modulation of GSK3β autoinhibition by thr-7 and thr-8[J]. FEBS Lett, 2018, 592(4): 537-546. [21] WANG Z, ZHAN Y, XU J, et al. β-Sitosterol reverses multidrug resistance via BCRP suppression by inhibiting the p53-MDM2 interaction in colorectal cancer[J]. J Agric Food Chem, 2020, 68(12): 3850-3858. [22] CHENG C, DONG W. Aloe-Emodin induces endoplasmic reticulum stress-dependent apoptosis in colorectal cancer cells[J]. Med Sci Monit, 2018, 24: 6331-6339. [23] RU JL, LI P, WANG JN, et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines[J]. J Cheminform, 2014, 6: 13. -





 下载:
下载: