Huangqi-Danshen in the Regulation of ASIC1a/CaMKⅡδ Signaling to Inhibit Cardiomyocytes from Pyroptosis in Acidic Microenvironment
-
摘要:
目的 探讨黄芪丹参配伍对乳酸诱导的酸性微环境中心肌细胞焦亡损伤的保护作用及基于酸敏感离子通道1a(ASIC1a)/钙离子-钙调蛋白依赖性激酶Ⅱδ(CaMKⅡδ)信号通路的调控机制。 方法 采用Lactate刺激H9C2心肌细胞建立pH 6.5酸性微环境细胞损伤模型。分为正常对照组、模型组、黄芪丹参提取物(HQ)组、HQ+ASIC激动剂Nocistatin组和ASIC抑制剂NS383阳性药对照组。采用CCK-8法测定心肌细胞活力, 流式细胞术检测心肌细胞凋亡, 免疫荧光检测ASIC1a表达, qPCR分析NLRP3、Caspase-1、IL-1β及GSDMD mRNA表达, Western blot检测ASIC1a、CaMKⅡδ、NLRP3、Caspase-1、Cleaved Caspase-1、GSDMD及GSDME-N蛋白的表达。 结果 在pH 6.5酸性环境中, 心肌细胞活力显著降低(P < 0.01), 细胞凋亡明显增加(P < 0.01), NLRP3、Caspase-1、IL-1β及GSDMD mRNA表达明显增加(P < 0.01), ASIC1a、CaMKⅡδ、NLRP3、Caspase-1、Cleaved Caspase-1、GSDMD及GSDME-N蛋白表达明显增加(P < 0.01)。60 μg · mL-1HQ可显著促进pH6.5酸性微环境中心肌细胞活力(P < 0.01), 抑制心肌细胞凋亡(P < 0.01), 下调NLRP3、Caspase-1、IL-1β、GSDMD mRNA和ASIC1a、CaMKⅡδ、NLRP3、Caspase-1、Cleaved Caspase-1、GSDMD及GSDME-N蛋白表达(P < 0.01)。并且HQ的以上效应可被ASIC激动剂Nocistatin逆转。 结论 黄芪丹参配伍可下调ASIC1a/CaMKⅡδ信号抑制酸性微环境下NLRP3炎症小体介导的心肌细胞焦亡。 Abstract:OBJECTIVE To observe the protective effect of Huangqi-Danshen on cardiomyocytes pyroptosis or injury in acidic microenvironment induced by lactic acid and the regulatory mechanism based on the acid-sensitive ion channel 1a (ASIC1a) or calcium/calmodulin-dependent protein kinase Ⅱδ (Ca MKⅡδ) signaling pathway. METHODS Lactate was used to stimulate H9C2 cardiomyocytes to establish the cell injury model in the acidic environment of pH 6.5. The rats were divided into the normal control group, model group, Huangqi-Danshen extract (HQ) group, HQ + ASIC agonist Nocistatin group, as well as ASIC inhibitor NS383 positive drug control group. The viability of cardiomyocytes was measured by CCK-8 method. The apoptosis of cardiomyocytes was detected by flow cytometry. Besides, the expression of ASIC1a was detected by immunofluorescence. The mRNA expression of NLRP3, Caspase-1, IL-1β, and GSDMD was analyzed by qPCR. In addition, the protein expression of ASIC1a, CaMKⅡδ, NLRP3, Caspase-1, Cleaved Caspase-1, GSDMD, and GSDME-N was detected by Western blot. RESULTS In the acidic environment of pH 6.5, the viability of cardiomyocytes was significantly reduced (P < 0.01), but the apoptosis was significantly increased (P < 0.01). The mRNA expressions of NLRP3, Caspase-1, IL-1β and GSDMD were significantly increased (P < 0.01), and the protein expressions of ASIC1a, CaMKⅡδ, NLRP3, Caspase-1, Cleaved Caspase-1, GSDMD and GSDME-N were also significantly increased (P < 0.01). In addition, we found that HQ (60 μg·mL-1) could significantly promote the viability of cardiomyocytes in acidic microenvironment (P < 0.01), inhibit cardiomyocyte apoptosis (P < 0.01), and down-regulate the mRNA expressions (NLRP3, Caspase-1, IL-1β, GSDMD) combined with protein expressions (ASIC1a, CaMKⅡδ, NLRP3, Caspase-1, Cleaved Caspase-1, GSDMD, GSDME-N) (P < 0.01). Besides, the above effects of HQ can be reversed by ASIC agonist Nocistatin. CONCLUSION Huangqi-Danshen can down-regulate ASIC1a/CaMKⅡδ signaling to inhibit NLRP3 inflammasome -mediated cardiomyocyte pyroptosis in acidic microenvironment. -
Key words:
- Huangqi-Danshen /
- coronary heart disease /
- ASIC1a /
- acidic microenvironment /
- pyroptosis
-
图 1 CCK8检测细胞活力
]注: 与Blank组相比, * *P < 0.01;与Control组相比, ##P < 0.01; 与HQ 60 μg · mL-1组相比, ▲P < 0.05。x±s, n=6。]>
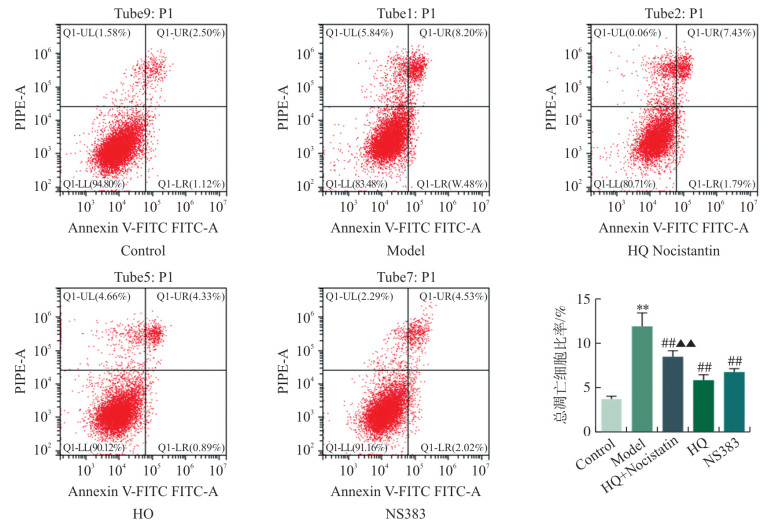
HQ抑制pH 6.5酸性环境中心肌细胞凋亡 本研究进一步采用流式细胞术检测pH 6.5酸性环境中心肌细胞凋亡, 结果显示: pH 6.5酸性环境可诱导细胞凋亡明显增加(P < 0.01), HQ和ASIC1a抑制剂NS383均可减少酸性环境中的心肌细胞凋亡(P < 0.01), 且HQ的这种保护效应可被ASIC1a激动剂Nocistatin逆转, 提示HQ对酸性环境中的心肌细胞凋亡的保护效应与调控ASIC1a酸敏感Ca2+通道有关(图 2)。 流式细胞术检测各组pH 6.5酸性环境中心肌细胞凋亡 Flow cytometry detection of cardiomyocyte apoptosis in the acidic environment of pH 6.5 in each group 注: Q1-UL.坏死细胞;Q1-UR.晚期凋亡细胞;Q1-LL.正常细胞;Q1-LR.早期凋亡细胞。与Control组相比, * *P < 0.01;与Model组相比, ##P < 0.01;与HQ组相比, ▲▲P < 0.01。x±s, n=3。 Figure 1. CCK8 assay for cell viability
图 5 pH 6.5酸性环境中心肌细胞ASIC1a、CaMKⅡδ、NLRP3、Caspase-1、Cleaved Caspase-1、GSDMD及GSDME-N蛋白表达比较
注: 与Control组相比, * *P < 0.01;与Model组相比, #P < 0.05, ##P < 0.01;与HQ组相比, ▲P < 0.05, ▲▲P < 0.01。x±s, n=3。
Figure 5. Comparison of cardiomyocytes protein expression of ASIC1a, CaMKⅡδ, NLRP3, Caspase-1, Cleaved Caspase-1, GSDMD and GSDME-N in the acidic environment of pH 6.5
表 1 特定引物序列
Table 1. Specific primer sequences
引物 序列(5′→3′) NLRP3 Forward
Reverse5′-GTGTTTCGAATCCCACTGTG-3′
5′-TCTGCTTCTCACGTACTTTCTG-3′Caspase-1 Forward
Reverse5′-ACGCCTTGCCCTCATAAT-3′
5′-TCTAATACATCTGGGACTTCTT-3′IL-1β Forward
Reverse5′-ATGCACCTGTACGATCACTG-3′
5′-ACAAAGGACATGGAGAACACC-3′GSDMD Forward
Reverse5′-CTACCTTACCTCATGCCGTTCGT-3′
5′-ACCTCCGTCCGAGAAGTCTGAA-3′β-actin Forward
Reverse5′-TACAACTCCTTGCAGCTCC-3′
5′-ATCTTCATGAGGTAGTCAGTC-3′ -
[1] SANSBURY BE, DEMARTINO AM, XIE ZZ, et al. Metabolomic analysis of pressure-overloaded and infarcted mouse hearts[J]. Circ Heart Fail, 2014, 7(4): 634-642. doi: 10.1161/CIRCHEARTFAILURE.114.001151 [2] REDD MA, SCHEUER SE, SAEZ NJ, et al. Therapeutic inhibition of acid-sensing ion channel 1a recovers heart function after ischemia-reperfusion injury[J]. Circulation, 2021, 144(12): 947-960. doi: 10.1161/CIRCULATIONAHA.121.054360 [3] TREVANI AS, ANDONEGUI G, GIORDANO M, et al. Extracellular acidification induces human neutrophil activation[J]. J Immunol, 1999, 162(8): 4849-4857. doi: 10.4049/jimmunol.162.8.4849 [4] WALDMANN R, CHAMPIGNY G, BASSILANA F, et al. A proton-gated cation channel involved in acid-sensing[J]. Nature, 1997, 386(6621): 173-177. doi: 10.1038/386173a0 [5] YODER N, YOSHIOKA C, GOUAUX E. Gating mechanisms of acid-sensing ion channels[J]. Nature, 2018, 555(7696): 397-401. doi: 10.1038/nature25782 [6] FRIESE MA, CRANER MJ, ETZENSPERGER R, et al. Acid-sensing ion channel-1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system[J]. Nat Med, 2007, 13(12): 1483-1489. doi: 10.1038/nm1668 [7] LYNAGH T, ROMERO-ROJO JL, LUND C, et al. Molecular basis for allosteric inhibition of acid-sensing ion channel 1a by ibuprofen[J]. J Med Chem, 2017, 60(19): 8192-8200. doi: 10.1021/acs.jmedchem.7b01072 [8] YANG YJ, JIANG K, LIU X, et al. CaMKⅡ in regulation of cell death during myocardial reperfusion injury[J]. Front Mol Biosci, 2021, 8: 668129. doi: 10.3389/fmolb.2021.668129 [9] WILLEFORD A, SUETOMI T, NICKLE A, et al. CaMKⅡδ-mediated inflammatory gene expression and inflammasome activation in cardiomyocytes initiate inflammation and induce fibrosis[J]. JCI Insight, 2018, 3(12): e97054. doi: 10.1172/jci.insight.97054 [10] 祁晓霞, 董宇, 单晨啸, 等. 基于UFLC-Q-TOF/MS分析黄芪-丹参药对化学成分研究[J]. 南京中医药大学学报, 2017, 33(1): 93-96, 103. https://www.cnki.com.cn/Article/CJFDTOTAL-NJZY201701022.htmQI XX, DONG Y, SHAN CX, et al. Analysis of the main components in herbal pair: Astragali Radix and salviae miltiorrhizae by UFLC-Q-TOF/MS[J]. J Nanjing Univ Tradit Chin Med, 2017, 33(1): 93-96, 103. https://www.cnki.com.cn/Article/CJFDTOTAL-NJZY201701022.htm [11] 王新东, 祁晓霞, 卞勇, 等. 黄芪丹参水煎液抑制ISO诱导的大鼠心肌重构及其下调STIM1, TRPC1, CaN和NFATc3表达的机制[J]. 中国中药杂志, 2017, 42(14): 2738-2743. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201714020.htmWANG XD, QI XX, BIAN Y, et al. Huangqi Danshen Decoction attenuates isoproterenol-induced myocardial remodeling via STIM1, TRPC1, CaN and NFATc3 pathways in rats[J]. China J Chin Mater Med, 2017, 42(14): 2738-2743. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201714020.htm [12] 王新东, 卞勇, 祁晓霞. 黄芪丹参水煎液激活AMPK上调自噬抑制ISO诱导的大鼠心肌重构[J]. 中药材, 2017, 40(10): 2433-2436. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYCA201710045.htmWANG XD, BIAN Y, QI XX. The decoction of Radix Astragali and Radix Salviae Miltiorrhizae activates AMPK, upregulates autophagy and inhibits ISO-induced myocardial remodeling in rats[J]. J Chin Med Mater, 2017, 40(10): 2433-2436. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYCA201710045.htm [13] 张蒙, 王新东. 黄芪甲苷对缺氧/复氧诱导大鼠心肌细胞能量代谢重编程的影响及机制[J]. 山东医药, 2021, 61(31): 13-16. https://www.cnki.com.cn/Article/CJFDTOTAL-SDYY202131007.htmZHANG M, WANG XD. Effect and mechanism of astragaloside Ⅳ on energy metabolism reprogramming induced by hypoxia/reoxygenation in rat cardiomyocytes[J]. Shandong Med J, 2021, 61(31): 13-16. https://www.cnki.com.cn/Article/CJFDTOTAL-SDYY202131007.htm [14] TAN Z, XIE N, BANERJEE S, et al. The monocarboxylate transporter 4 is required for glycolytic reprogramming and inflammatory response in macrophages[J]. J Biol Chem, 2015, 290(1): 46-55. doi: 10.1074/jbc.M114.603589 [15] LIN HC, CHEN YJ, WEI YH, et al. Lactic acid fermentation is required for NLRP3 inflammasome activation[J]. Front Immunol, 2021, 12: 630380. doi: 10.3389/fimmu.2021.630380 [16] SUETOMI T, MIYAMOTO S, BROWN JH. Inflammation in nonischemic heart disease: Initiation by cardiomyocyte CaMKⅡ and NLRP3 inflammasome signaling[J]. Am J Physiol Heart Circ Physiol, 2019, 317(5): H877-H890. doi: 10.1152/ajpheart.00223.2019 [17] FENG N, ANDERSON ME. CaMKⅡ is a nodal signal for multiple programmed cell death pathways in heart[J]. J Mol Cell Cardiol, 2017, 103: 102-109. doi: 10.1016/j.yjmcc.2016.12.007 [18] LUCZAK ED, WU YJ, GRANGER JM, et al. Mitochondrial CaMKⅡ causes adverse metabolic reprogramming and dilated cardiomyopathy[J]. Nat Commun, 2020, 11(1): 4416. doi: 10.1038/s41467-020-18165-6 [19] JIANG HL, XING JG, FANG JS, et al. Tilianin protects against ischemia/reperfusion-induced myocardial injury through the inhibition of the Ca2+/calmodulin-dependent protein kinase Ⅱ-dependent apoptotic and inflammatory signaling pathways[J]. Biomed Res Int, 2020, 2020: 5939715. -





 下载:
下载: