Establishment of HPLC Characteristic Chromatogram and Study on Quality Value Transmitting of Substance Benchmarks of Yihuang Decoction
-
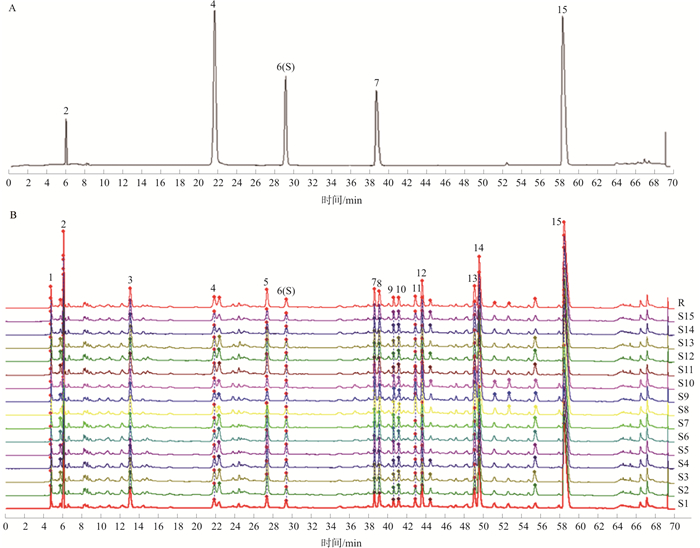
摘要:
目的 建立易黄汤物质基准的HPLC特征图谱和易黄汤中指标性成分尿囊素、没食子酸、京尼平苷酸和小檗碱的含量测定方法, 阐明这4种成分在饮片-水煎液-物质基准的量值传递规律。 方法 采用Agilent ZORBAX SB-Aq色谱柱(250 mm×4.6 mm, 5 μm), 以乙腈-0.1%乙酸水溶液为流动相, 梯度洗脱, 采用多波长切换法, 检测波长分别为205、254、280 nm, 以上述4种成分的转移率为主要评价指标, 研究从饮片-水煎液-物质基准的量值传递规律。 结果 建立了易黄汤物质基准的HPLC特征图谱, 15批物质基准冻干粉样品相似度均大于0.90, 共标定出15个共有峰, 指认出其中5个特征峰(2号峰尿囊素、4号峰没食子酸、6号峰京尼平苷酸、7号峰黄柏碱、15号峰小檗碱); 同时进行含量测定, 15批物质基准中尿囊素、没食子酸、京尼平苷酸、小檗碱的含量范围分别为0.801%~1.087%、0.047%~0.057%、0.037%~0.081%、0.250%~0.387%;15批易黄汤水煎液到物质基准的量值传递(转移率)分别为尿囊素73.09%~91.37%、没食子酸74.99%~89.32%、京尼平苷酸71.77%~94.38%、小檗碱76.54%~88.43%。 结论 建立的特征图谱和4种成分的含量测定方法能较好地控制易黄汤物质基准的质量, 量值传递结果表明物质基准冻干粉的制备工艺具有科学性和稳定性, 为经典名方易黄汤制剂的后续开发打下了基础。 Abstract:OBJECTIVE To establish the HPLC characteristic chromatogram, simultaneously determinate the contents of allantoin, gallic acid, geniposidic acid and berberine in the substance benchmark of Yihuang Decoction, and clarify the quality value transmitting rules from decoction pieces to water decoction to substance benchmarks. METHODS Agilent ZORBAX SB-Aq chromatographic column (250 mm×4.6 mm, 5 μm) was used, eluted with acetonitrile-0.1% acetic acid in gradient elution, and the detection wavelength was 205, 254, and 280 nm, respectively. The transfer rates of the above four components were used as the indexes for quality evaluation to study the quality value transmitting rule from decoction pieces to water decoction to substance benchmarks. RESULTS The HPLC characteristic chromatogram of Yihuang Decoction was established, the similarity of 15 batches of samples was greater than 0.90, and a total of 15 common peaks were found and five of them [allantoin (peak 2), gallic acid (peak 4), geniposidic acid (peak 6), phellodendrine (peak 7), berberine (peak 15)] were identified. The contents of allantoin, gallic acid, geniposidic acid and berberine in 15 batches of benchmarks were 0.801%-1.087%, 0.047%-0.057%, 0.037%-0.081%, 0.250%-0.387%, respectively. The transfer rates (from water decoction to substance benchmarks) of allantoin, gallic acid, geniposidic acid and berberine were 73.09%-91.37%, 74.99%-89.32%, 71.77%-94.38%, 76.54%-88.43%, respectively. CONCLUSION The HPLC characteristic chromatogram combined with the analytical method of multicomponents in simultaneous quantitation established in this experiment are stable and reliable, which can provide a theoretical guidance for the quality evaluation of Yihuang Decoction. The quality value transmitting results shows that the preparing technology of substance benchmarks is scientific and stable, which can lay a foundation for the subsequent development of classic prescription Yihuang Decoction preparation. -
表 1 易黄汤饮片来源
Table 1. Sources of Yihuang Decoction pieces
名称 炮制工艺 批号 产地 炒山药 取净山药片, 置热锅中, 140 ℃炒制约60 min, 炒至表面微黄色, 取出, 晾凉。 2021110401~2021110404
2021110501~2021110506河南温县
河南武陟炒芡实 取净芡实, 置热锅中, 160 ℃炒制约70 min, 炒至表面微黄色, 取出, 晾凉。 2021083101~2021083106
2021091101~2021091104江苏高邮 炒白果 取净白果仁, 置热锅中, 180 ℃炒制约15 min, 炒至表面色泽加深, 略有焦斑, 横断面胶质样, 外层黄色, 内层淡黄色, 粉性, 中间有空隙。有香气。 2021081001~2021081010 江苏邳州 酒车前子 取净车前子, 喷淋定量黄酒拌匀, 闷润0.5 h, 待酒被吸尽后, 180 ℃炒至爆裂声起, 略有香气逸出时, 取出晾凉。每100 kg车前子, 用黄酒20 kg。 2021063001~2021063004
2021070101~2021070106江西新干 盐黄柏 取原药材, 除去杂质, 洗净, 加水喷淋(水量为药材质量的15%), 闷润约45 min至内外湿度一致, 切丝(1 cm), 70 ℃烘30 min, 至干。取净黄柏丝, 加30%药材量的水配制食盐水, 闷润约0.5 h, 待盐水被吸尽后, 置热锅中, 160 ℃炒制约40 min, 至干。每100 kg黄柏, 用食盐2 kg。 2021073101~2021073105
2021080101~2021080105四川大邑
四川雅安表 2 易黄汤15批物质基准对应的饮片批次
Table 2. 15 batches of Yihuang Decoction substance benchmark corresponding to the batches of decoction pieces
批号 炒山药 炒芡实 炒白果 盐黄柏 酒车前子 S1 2021110402 2021083105 2021081001 2021073103 2021070101 S2 2021110501 2021083103 2021081004 2021073104 2021063001 S3 2021110403 2021083104 2021081009 2021073105 2021063004 S4 2021110502 2021091104 2021081006 2021080101 2021063002 S5 2021110404 2021091103 2021081004 2021080102 2021063003 S6 2021110503 2021091101 2021081005 2021073103 2021063001 S7 2021110403 2021091102 2021081008 2021073104 2021063004 S8 2021110501 2021083103 2021081007 2021073102 2021070106 S9 2021110502 2021083101 2021081002 2021073103 2021070105 S10 2021110401 2021083106 2021081005 2021073101 2021070103 S11 2021110506 2021083101 2021081004 2021073104 2021070102 S12 2021110402 2021083104 2021081010 2021080104 2021070104 S13 2021110504 2021091103 2021081009 2021080105 2021070102 S14 2021110505 2021083103 2021081002 2021080103 2021070105 S15 2021110502 2021083102 2021081003 2021073102 2021070106 表 3 易黄汤15批物质基准特征图谱的相似度
Table 3. The similarity of 15 batches of Yihuang Decoction substance benchmark
S1 S2 S3 S4 S5 S6 S7 S8 S9 S10 S11 S12 S13 S14 S15 对照 S1 1 0.984 0.985 0.985 0.985 0.985 0.985 0.985 0.984 0.985 0.986 0.986 0.984 1 1 0.985 S2 1 0.999 0.999 0.996 0.996 0.996 0.968 0.968 0.966 0.999 0.999 1 0.984 0.985 0.986 S3 1 1 0.995 0.995 0.995 0.967 0.967 0.967 1 1 0.999 0.985 0.985 0.975 S4 1 0.995 0.995 0.995 0.967 0.967 0.967 1 1 0.999 0.985 0.985 0.964 S5 1 1 1 0.972 0.972 0.971 0.995 0.995 0.997 0.985 0.985 0.997 S6 1 1 0.972 0.972 0.97 0.996 0.996 0.997 0.985 0.985 0.989 S7 1 0.972 0.972 0.971 0.995 0.995 0.996 0.985 0.985 0.998 S8 1 0.999 0.999 0.968 0.968 0.968 0.983 0.984 0.977 S9 1 0.999 0.968 0.968 0.968 0.983 0.984 0.986 S10 1 0.968 0.968 0.967 0.984 0.984 0.988 S11 1 1 0.999 0.985 0.986 0.985 S12 1 0.999 0.986 0.986 0.997 S13 1 0.984 0.984 0.99 S14 1 1 0.968 S15 1 0.998 表 4 含量测定方法学考察结果(%,n=6)
Table 4. Assay methodology (%, n=6)
指标性成分 回归方程 R2 RSD 平均加样回收率 精密度 重复性 稳定性 尿囊素 y=22 793x+164 611 0.999 1 1.4 0.9 0.9 108.23 没食子酸 y=59 952x-139 067 0.999 8 1.0 1.2 1.5 104.60 京尼平苷酸 y=13 829x-15 788 0.999 9 0.6 0.7 0.7 103.08 小檗碱 y=60 255x-49 006 0.999 9 0.3 0.9 0.7 99.69 注: 专属性均良好。 表 5 饮片中指标性成分的含量(%, n=2)
Table 5. Content of index components in decoction pieces (%, n=2)
批次 尿囊素 没食子酸 京尼平苷酸 小檗碱 S1 0.631 0.033 1.333 4.928 S2 0.621 0.034 1.226 5.439 S3 0.543 0.031 1.273 4.952 S4 0.558 0.028 1.271 4.883 S5 0.574 0.029 1.225 4.900 S6 0.495 0.034 1.226 4.928 S7 0.543 0.027 1.273 5.439 S8 0.621 0.034 1.273 5.022 S9 0.548 0.034 1.327 4.928 S10 0.463 0.033 1.099 5.434 S11 0.462 0.034 1.100 4.928 S12 0.495 0.031 1.321 5.320 S13 0.574 0.029 1.100 4.919 S14 0.621 0.034 1.327 5.405 S15 0.548 0.026 1.273 5.029 x±s 0.553±0.056 0.031±0.003 1.243±0.082 5.097±0.232 表 6 水煎液中指标性成分的含量及出膏率(%, n=2)
Table 6. Content of index components and extraction rate in Decoction (%, n=2)
批次 水煎液中含量 出膏率 尿囊素 没食子酸 京尼平苷酸 小檗碱 S1 0.187 7 0.008 0 0.012 8 0.054 2 13.94 S2 0.181 2 0.008 0 0.012 9 0.054 4 14.64 S3 0.173 5 0.007 8 0.012 1 0.052 8 14.70 S4 0.156 2 0.009 1 0.007 1 0.045 2 14.33 S5 0.160 6 0.009 1 0.005 6 0.040 1 15.14 S6 0.151 7 0.009 1 0.005 5 0.040 2 14.04 S7 0.168 8 0.008 4 0.008 9 0.054 9 13.19 S8 0.170 0 0.008 4 0.009 1 0.054 8 12.49 S9 0.177 4 0.008 4 0.009 2 0.054 8 12.84 S10 0.158 4 0.008 0 0.010 8 0.056 7 14.90 S11 0.158 7 0.008 0 0.011 0 0.056 8 16.37 S12 0.157 0 0.008 0 0.010 9 0.056 7 15.16 S13 0.143 3 0.007 8 0.010 7 0.064 1 16.29 S14 0.150 8 0.007 8 0.010 8 0.064 0 15.63 S15 0.145 2 0.007 8 0.010 8 0.064 2 14.77 x±s 0.163±0.013 0.008±0.001 0.010±0.002 0.054±0.008 14.560±1.138 表 7 物质基准中指标性成分的含量及干粉得率(%, n=2)
Table 7. Content of index components and dry powder yield in substance standard (%, n=2)
批次 物质基准中含量 干粉得率 尿囊素 没食子酸 京尼平苷酸 小檗碱 S1 1.087 0.049 0.080 0.338 12.89 S2 1.072 0.050 0.081 0.339 13.20 S3 1.079 0.050 0.081 0.339 13.07 S4 0.958 0.057 0.037 0.250 13.79 S5 0.999 0.057 0.037 0.250 13.44 S6 1.003 0.057 0.038 0.254 13.30 S7 1.068 0.051 0.057 0.353 12.36 S8 1.058 0.052 0.058 0.355 12.30 S9 1.054 0.051 0.058 0.352 12.42 S10 1.001 0.052 0.075 0.365 13.66 S11 0.944 0.052 0.075 0.366 13.54 S12 0.992 0.052 0.076 0.367 13.50 S13 0.865 0.047 0.066 0.383 14.61 S14 0.850 0.048 0.067 0.387 14.62 S15 0.801 0.047 0.066 0.384 14.66 x±s 0.989±0.090 0.051±0.003 0.063±0.016 0.339±0.048 13.420±0.777 表 8 饮片到水煎液中指标性成分的转移率(%, n=2)
Table 8. Transfer rate of index components from decoction pieces to Decoction (%, n=2)
批次 尿囊素 没食子酸 京尼平苷酸 小檗碱 S1 76.37 61.26 24.75 14.13 S2 74.89 60.73 26.98 12.84 S3 82.04 65.05 24.34 13.68 S4 71.82 84.16 14.27 11.88 S5 71.87 81.05 11.73 10.50 S6 78.69 69.02 11.54 10.46 S7 79.85 80.97 18.00 12.96 S8 70.25 63.93 18.27 14.01 S9 83.05 63.68 17.79 14.27 S10 87.76 63.03 25.15 13.39 S11 88.17 61.20 25.60 14.81 S12 81.48 67.32 21.22 13.70 S13 64.16 69.52 24.96 16.73 S14 62.34 59.73 20.85 15.20 S15 67.99 78.14 21.74 16.39 x±s 76.05±7.95 68.59±8.38 20.48±5.06 13.66±1.81 表 9 水煎液到物质基准中指标性成分的转移率及浸膏转移率(%, n=2)
Table 9. Transfer rate of index components and extract from Decoction to substance standard (%, n=2)
批次 水煎液到物质基准指标成分转移率 浸膏转移率 尿囊素 没食子酸 京尼平苷酸 小檗碱 S1 74.61 79.27 80.71 80.40 92.44 S2 76.28 81.15 81.16 80.27 90.15 S3 80.16 83.60 86.12 82.74 88.88 S4 84.69 87.05 71.77 76.54 96.24 S5 85.90 86.37 90.52 86.29 88.81 S6 91.37 87.46 94.38 87.41 94.72 S7 77.79 74.99 78.82 78.99 93.71 S8 76.54 75.57 78.75 79.62 98.52 S9 73.09 75.28 77.36 79.11 96.75 S10 85.59 88.30 93.97 87.06 91.65 S11 80.52 88.42 92.68 87.12 82.71 S12 85.51 88.12 93.82 87.67 89.05 S13 88.20 89.27 90.96 87.27 89.72 S14 82.44 89.32 90.43 88.43 93.53 S15 80.72 88.24 89.06 87.51 99.24 x±s 81.56±5.30 84.16±5.44 86.03±7.30 83.76±4.17 92.41±4.39 -
[1] 傅山. 傅青主女科[M]. 北京: 中国中医药出版社, 2019: 5.FU S. Fu Qingzhu's Obstetrics and Gynecologyis[M]. Beijing: China press of traditional Chinese medicine, 2019: 5. [2] 刘春生. 《傅青主女科》医方集解系列(Ⅳ) 黄带下[J]. 中国中医药现代远程教育, 2017, 15(4): 63-65. doi: 10.3969/j.issn.1672-2779.2017.04.026LIU CS. Collected exegesis of Recipes of Fu Qingzhu's Obstetrics and Gynecology (Ⅳ) Yellow vaginal discharge[J]. Chin Med Mod Distance Educ China, 2017, 15(4): 63-65. doi: 10.3969/j.issn.1672-2779.2017.04.026 [3] 许桢惠. 加减易黄汤联合保妇康栓治疗老年阴道炎临床观察[J]. 光明中医, 2019, 34(7): 984-986. doi: 10.3969/j.issn.1003-8914.2019.07.006XU ZH. Clinical observation on modified Yihuang Decoction combined with baofukang suppository in the treatment of senile vaginitis[J]. Guangming J Chin Med, 2019, 34(7): 984-986. doi: 10.3969/j.issn.1003-8914.2019.07.006 [4] 王艳春. 易黄汤加减治疗脾虚湿热型阴道炎的临床疗效研究[J]. 中医临床研究, 2021, 13(12): 81-83. doi: 10.3969/j.issn.1674-7860.2021.12.029WANG YC. Study on the clinical effect of modified Yihuang Decoction on vaginitis of dampness-heat type with spleen deficiency[J]. Clin J Chin Med, 2021, 13(12): 81-83. doi: 10.3969/j.issn.1674-7860.2021.12.029 [5] 王秀清, 刘宇灵, 林龙飞, 等. 易黄汤阴道用温敏原位凝胶及其普通凝胶的流变学和体外释放研究[J]. 中国中药杂志, 2020, 45(3): 539-547. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202003013.htmWANG XQ, LIU YL, LIN LF, et al. Rheology and in vitro release properties of thermosensitive in situ gel of Yihuang Decoction and its common gel for vaginal use[J]. China J Chin Mater Med, 2020, 45(3): 539-547. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY202003013.htm [6] 武雪莲. 加味易黄汤联合自制清毒方对宫颈HPV病毒感染患者病毒载量及TNF-α、hs-CRP水平的影响[J]. 光明中医, 2021, 36(1): 84-86. doi: 10.3969/j.issn.1003-8914.2021.01.032WU XL. Effect of oral administration of Yihuang Decoction and self-made qingdu prescription on viral load, TNF-α and hs-CRP level in patients with cervical HPV infection[J]. Guangming J Chin Med, 2021, 36(1): 84-86. doi: 10.3969/j.issn.1003-8914.2021.01.032 [7] 国家药品监督管理局. 古代经典名方中药复方制剂简化注册审批管理规定[EB/OL]. (2018-05-29)[2022-01-16]. https://www.nmpa.gov.cn/zhuanti/ypqxgg/ggzhcfg/20180601163901361.html.National Medical Products Administration. The simplified registration approval management of ancient classic prescriptions of traditional Chinese medicine compound preparations[EB/OL]. (2018-05-29)[2022-01-16]. https://www.nmpa.gov.cn/zhuanti/ypqxgg/ggzhcfg/20180601163901361.html. [8] 付媛媛, 蒋玉兰, 单鸣秋, 等. 盐黄柏饮片与易黄汤的特征图谱与主要成分测定研究[J]. 中草药, 2020, 51(10): 2790-2797. doi: 10.7501/j.issn.0253-2670.2020.10.021FU YY, JIANG YL, SHAN MQ, et al. Study on characteristic chromatogram and determination of indicative components of salt-fried Phellodendri chinensis cortex and Yihuang Decoction[J]. Chin Tradit Herb Drugs, 2020, 51(10): 2790-2797. doi: 10.7501/j.issn.0253-2670.2020.10.021 [9] 毕嘉谣, 田湾湾, 张翼, 等. 经典名方易黄汤物质基准的量值传递分析[J]. 中国实验方剂学杂志, 2021, 27(16): 24-31. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX202116004.htmBI JY, TIAN WW, ZHANG Y, et al. Analysis on quality transmitting of substance benchmarks of yihuangtang[J]. Chin J Exp Tradit Med Formulae, 2021, 27(16): 24-31. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX202116004.htm [10] 田湾湾, 钟琳瑛, 张琦, 等. 车前子酒炙工艺优选及其不同炮制品对比研究[J]. 天津中医药, 2020, 37(2): 220-226. https://www.cnki.com.cn/Article/CJFDTOTAL-TJZY202002026.htmTIAN WW, ZHONG LY, ZHANG Q, et al. Optimum processing technology with wine and comparative study of different processed products of Plantaginis Semen[J]. Tianjin J Tradit Chin Med, 2020, 37(2): 220-226. https://www.cnki.com.cn/Article/CJFDTOTAL-TJZY202002026.htm [11] 国家药典委员会. 中华人民共和国药典: 一部[M]. 北京: 中国医药科技出版社, 2020.National Pharmacopoeia Commission. Pharmacopoeia of People's Republic of China: Ⅰ[M]. Beijing: China medical science press, 2020. [12] 田伟, 甄亚钦, 董秋菊, 等. 车前子煎煮过程中4种化学成分含量变化规律研究[J]. 中国新药杂志, 2018, 27(16): 1927-1931. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXYZ201816018.htmTIAN W, ZHEN YQ, DONG QJ, et al. Variation of four chemical components in plantaginis semen during decoction process[J]. Chin J New Drugs, 2018, 27(16): 1927-1931. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXYZ201816018.htm [13] 李敏. 山药活性成分提取技术及药理功能的研究进展[J]. 南方农业学报, 2013, 44(7): 1184-1190. https://www.cnki.com.cn/Article/CJFDTOTAL-GXNY201307027.htmLI M. Overview on extractive techniques and pharmacological functions of active constituents in Rhizoma dioscoreae[J]. J South Agric, 2013, 44(7): 1184-1190. https://www.cnki.com.cn/Article/CJFDTOTAL-GXNY201307027.htm [14] 朱群英, 张亚锋, 苏超男, 等. HILIC-HPLC双波长法测定山药中尿囊素和腺苷的含量[J]. 西北药学杂志, 2018, 33(3): 334-336. https://www.cnki.com.cn/Article/CJFDTOTAL-XBYZ201803012.htmZHU QY, ZHANG YF, SU CN, et al. Determination of allantoin and adenosine in rhizoma dioscoreae by HILIC-double-wavelength HPLC[J]. Northwest Pharm J, 2018, 33(3): 334-336. https://www.cnki.com.cn/Article/CJFDTOTAL-XBYZ201803012.htm [15] 王莹, 陈雪珊, 施洋, 等. RP-HPLC法测定山药中薯蓣皂苷的含量[J]. 新疆医科大学学报, 2017, 40(4): 516-517, 522. https://www.cnki.com.cn/Article/CJFDTOTAL-XJYY201704026.htmWANG Y, CHEN XS, SHI Y, et al. Determination of diosgenin in rhizoma dioscoreae by RP-HPLC[J]. J Xinjiang Med Univ, 2017, 40(4): 516-517, 522. https://www.cnki.com.cn/Article/CJFDTOTAL-XJYY201704026.htm [16] WANG XH, ZHAO HQ, MA CH, et al. Gallic acid attenuates allergic airway inflammation via suppressed interleukin-33 and group 2 innate lymphoid cells in ovalbumin-induced asthma in mice[J]. Int Forum Allergy Rhinol, 2018, 8(11): 1284-1290. [17] MARIN M, MARIA GINER R, RIOS JL, et al. Intestinal anti-inflammatory activity of ellagic acid in the acute and chronic dextrane sulfate sodium models of mice colitis[J]. J Ethnopharmacol, 2013, 150(3): 925-934. [18] 李冲冲, 龚苏晓, 许浚, 等. 车前子化学成分与药理作用研究进展及质量标志物预测分析[J]. 中草药, 2018, 49(6): 1233-1246. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201806001.htmLI CC, GONG SX, XU J, et al. Research progress on chemical composition and pharmacological effects of Semen Plantaginis and predictive analysis on Q-marker[J]. Chin Tradit Herb Drugs, 2018, 49(6): 1233-1246. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201806001.htm [19] 张慧娟, 李菊, 马晓慧, 等. 裂环环烯醚萜苷类化合物的药理作用研究进展[J]. 药学研究, 2018, 37(11): 659-663. https://www.cnki.com.cn/Article/CJFDTOTAL-SDYG201811012.htmZHANG HJ, LI J, MA XH, et al. Research progress on pharmacological effects of secoiridoid glycosides[J]. J Pharm Res, 2018, 37(11): 659-663. https://www.cnki.com.cn/Article/CJFDTOTAL-SDYG201811012.htm [20] BAE J, LEE DB, KIM YK, et al. Berberine protects 6-hydroxydopamine-induced human dopaminergic neuronal cell death through the induction of heme oxygenase-1[J]. Mol Cells, 2013, 35(2): 151-157. [21] KETTMANN V, KOSTALOVA D, HOLTJE HD. Human topoisomerase I poisoning: Docking protoberberines into a structure-based binding site model[J]. J Comput Aided Mol Des, 2004, 18(12): 785-796. -





 下载:
下载: