Huangqi Decoction Regulates TLR4/NF-κB Pathway to Improve High Glucose-Induced Podocyte Injury
-
摘要:
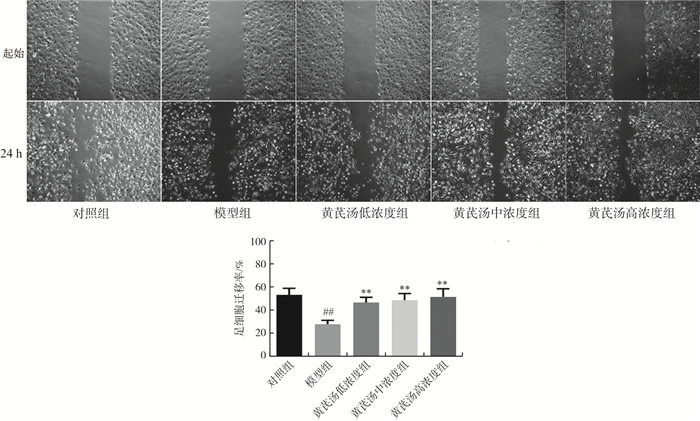
目的 探讨黄芪汤对高糖诱导足细胞损伤的作用及机制。 方法 体外培养人足细胞, 采用30 mmol·L-1葡萄糖干预24 h诱导足细胞损伤, 分别设置对照组、模型组、黄芪汤低浓度组(10 μg·mL-1)、黄芪汤中浓度组(30 μg·mL-1)和黄芪汤高浓度组(100 μg·mL-1)。采用CCK-8法检测细胞增殖能力, 划痕实验检测细胞迁移能力,qPCR法检测TNF-α、IL-6等炎症因子mRNA表达, ELISA法检测足细胞上清TNF-α、IL-6的含量,Western blot法检测足细胞TLR4、NF-κB、p-NF-κB、TNF-α及IL-6蛋白表达。 结果 与对照组比较, 模型组细胞划痕愈合率明显降低(P < 0.01),足细胞中TNF-α、IL-6、CCL24 mRNA表达水平,上清液中TNF-α、IL-6的含量和TLR4、p-NF-κB/NF-κB、TNF-α及IL-6蛋白表达均显著增加(P < 0.05,P < 0.01)。与模型组相比, 黄芪汤组细胞划痕率明显升高(P < 0.01),足细胞中TNF-α、IL-6的含量和mRNA表达,以及TLR4、p-NF-κB/NF-κB、TNF-α、IL-6蛋白表达均显著减少(P < 0.05, P < 0.01)。 结论 黄芪汤能有效减轻高糖对人足细胞的炎症损伤, 增强细胞增殖、迁移能力, 抑制细胞凋亡, 其机制可能与抑制TLR4/NF-κB信号通路、下调炎症因子的表达有关。 -
关键词:
- 黄芪汤 /
- 高糖 /
- 足细胞 /
- 炎症因子 /
- TLR4/NF-κB
Abstract:OBJECTIVE To investigate the effect and mechanism of Huangqi Decoction on high glucose-induced podocyte injury. METHODS Human podocytes were cultured in vitro and treated with 30 mmol·L-1 glucose for 24 h to induce injury. The groups were divided into control group, model group, low-, medium- and high-concentration of Huangqi Decoction groups (10, 30, 100 μg·mL-1). The cell proliferation was detected by CCK-8 method, the mRNA expression of TNF-α and IL-6 was detected by qPCR, the contents of TNF-α and IL-6 in podocyte supernatant were detected by ELISA, and the protein expression of TLR4, p-NF-κB, NF-κB, TNF-α and IL-6 in podocytes was detected by Western blot. RESULTS Compared with the control group, the mRNA expression of TNF-α, IL-6, CCL24, the contents of TNF-α and IL-6 and the protein expression of TLR4, p-NF-κB/NF-κB, TNF-α and IL-6 in podocytes significantly increased (P < 0.05, P < 0.01). Compared with the model group, the mRNA expression and contents of TNF-α, IL-6 and the protein expression of TLR4, p-NF-κB/NF-κB, TNF-α and IL-6 in podocytes of Huangqi Decoction groups significantly decreased (P < 0.05, P < 0.01). CONCLUSION Huangqi Decoction can reduce the inflammatory damage of human podocytes induced by high glucose, enhance cell proliferation and migration, and inhibit cell apoptosis. The mechanism may be related to the inhibition of TLR4/NF-κB signal pathway and down-regulation of the inflammatory factors expression. -
Key words:
- Huangqi Decoction /
- high glucose /
- podocyte /
- inflammatory factor /
- TLR4/NF-κB
-
表 1 细胞炎症因子引物序列
Table 1. Cellular inflammatory factor primer sequence
引物名称 上游引物 下游引物 β-actin 5'-GAAGTGTGACGTGGACATCC-3' 5'-CCGATCCACACGGAGTACTT-3' 分泌型磷蛋白1(SPP1) 5'-CTCCATTGACTCGAACGACTC-3' 5'-CAGGTCTGCGAAACTTCTTAGAT-3' IL-1α 5'-TGGTAGTAGCAACCAACGGGA-3' 5'-ACTTTGATTGAGGGCGTCATTC-3' IL-1β 5'-CCTGTCCTGCGTGTTGAAAGA-3' 5'-GGGAACTGGGCAGACTCAAA-3' IL-6 5'-ACTCACCTCTTCAGAACGAATTG-3' 5'-CCATCTTTGGAAGGTTCAGGTTG-3' IL-10 5'-TCAAGGCGCATGTGAACTCC-3' 5'-GATGTCAAACTCACTCATGGCT-3' IL-11 5'-CTGGGCTAGGGCATGAACTG-3' 5'-CTGGGACTCCAAGTGCAAGA-3' TNF-α 5'-CCCAGGCAGTCAGATCATCTTC-3' 5'-GCTGCCCCTCAGCTTGAG-3' CC趋化因子配体(CCL)2 5'-CAGCCAGATGCAATCAATGCC-3' 5'-TGGAATCCTGAACCCACTTCT-3' CCL3 5'-AGTTCTCTGCATCACTTGCTG-3' 5'-CGGCTTCGCTTGGTTAGGAA-3' CCL8 5'-TGGAGAGCTACACAAGAATCACC-3' 5'-TGGTCCAGATGCTTCATGGAA-3' CCL20 5'-TGCTGTACCAAGAGTTTGCTC-3' 5'-CGCACACAGACAACTTTTTCTTT-3' CCL24 5'-ACATCATCCCTACGGGCTCT-3' 5'-CTTGGGGTCGCCACAGAAC-3' CXC趋化因子受体(CXCR)1 5'-CTGACCCAGAAGCGTCACTTG-3' 5'-CCAGGACCTCATAGCAAACTG-3' CXCR5 5'-CACGTTGCACCTTCTCCCAA-3' 5'-GGAATCCCGCCACATGGTAG-3' 表 2 不同浓度黄芪汤对足细胞增殖抑制率的影响(x±s, %, n=3)
Table 2. Effect of different concentration of Huangqi Decoction on podocyte proliferation (x±s, %, n=3)
黄芪汤/(μg·mL-1) 24 h 48 h 72 h 1 7.56±2.69 7.56±2.12 8.33±1.51 3 7.34±3.93 8.31±2.09 8.96±1.43 10 8.74±5.23 10.89±4.56 10.93±1.99 30 9.63±3.95 10.71±4.55 11.18±2.48 100 8.36±3.42 11.23±4.37 10.03±2.96 300 27.00±3.74** 45.62±6.31** 52.19±3.93** 1 000 42.08±3.18** 62.70±2.28** 66.92±6.02** 3 000 51.05±2.71** 78.48±1.05** 82.78±4.05** 注: 与黄芪汤1 μg·mL-1组比较, **P < 0.01。 -
[1] KALANTAR-ZADEH K, JAFAR TH, NITSCH D, et al. Chronic kidney disease[J]. Lancet, 2021, 398(10302): 786-802. doi: 10.1016/S0140-6736(21)00519-5 [2] CECCARELLI D, PALEARI R, SOLERTE B, et al. Re-thinking diabetic nephropathy: Microalbuminuria is just a piece of the diagnostic puzzle[J]. Clin Chim Acta, 2022, 524: 146-153. doi: 10.1016/j.cca.2021.11.009 [3] FOGO AB. Gains in understanding of podocyte loss[J]. Kidney Int, 2021, 100(5): 978-980. doi: 10.1016/j.kint.2021.08.003 [4] BARRERA-CHIMAL J, JAISSER F. Pathophysiologic mechanisms in diabetic kidney disease: A focus on current and future therapeutic targets[J]. Diabetes Obes Metab, 2020, 22(S1): 16-31. doi: 10.1111/dom.13969 [5] LI M, GUO QS, CAI HQ, et al. miR-218 regulates diabetic nephropathy via targeting IKK-β and modulating NK-κB-mediated inflammation[J]. J Cell Physiol, 2020, 235(4): 3362-3371. doi: 10.1002/jcp.29224 [6] LIN LY, LIN HF, WANG D, et al. Bone marrow mesenchymal stem cells ameliorated kidney fibrosis by attenuating TLR4/NF-κB in diabetic rats[J]. Life Sci, 2020, 262: 118385. doi: 10.1016/j.lfs.2020.118385 [7] 杨士瀛. 仁斋直指方论[M]. 福州: 福建科学技术出版社, 1989: 437.YANG SY. Ren-zhai's Direct Guidance on Formulas[M]. Fuzhou: Fujian science and technology press, 1989: 437. [8] ZHAO J, WANG L, CAO AL, et al. HuangQi Decoction ameliorates renal fibrosis via TGF-β/smad signaling pathway in vivo and in vitro[J]. Cell Physiol Biochem, 2016, 38(5): 1761-1774. doi: 10.1159/000443115 [9] GUO HJ, CAO AL, CHU S, et al. Astragaloside Ⅳ attenuates podocyte apoptosis mediated by endoplasmic Reticulum stress through upregulating sarco/endoplasmic Reticulum Ca2+-ATPase 2 expression in diabetic nephropathy[J]. Front Pharmacol, 2016, 7: 500. [10] LI ZZ, DENG WJ, CAO AL, et al. Huangqi Decoction inhibits hyperglycemia-induced podocyte apoptosis by down-regulated Nox4/p53/bax signaling in vitro and in vivo[J]. Am J Transl Res, 2019, 11(5): 3195-3212. [11] 陈骏良, 王利, 臧赢君, 等. 黄芪汤对肾脏集合管细胞ENaC活性的影响作用研究[J]. 上海医药, 2019, 40(3): 34-37. doi: 10.3969/j.issn.1006-1533.2019.03.014CHEN JL, WANG L, ZANG YJ, et al. Study on the effect of Huangqi Decoction on ENaC activity in renal collecting duct cell[J]. Shanghai Med Pharm J, 2019, 40(3): 34-37. doi: 10.3969/j.issn.1006-1533.2019.03.014 [12] CHU S, MAO XD, WANG L, et al. Effects of Huang qi decoction on endothelial dysfunction induced by homocysteine[J]. Evid Based Complement Alternat Med, 2016, 2016: 7272694. [13] CHEN F, ZHU XG, SUN ZQ, et al. Astilbin inhibits high glucose-induced inflammation and extracellular matrix accumulation by suppressing the TLR4/MyD88/NF-κB pathway in rat glomerular mesangial cells[J]. Front Pharmacol, 2018, 9: 1187. doi: 10.3389/fphar.2018.01187 [14] YANG XH, DANG XW, ZHANG X, et al. Liquiritin reduces lipopolysaccharide-aroused HaCaT cell inflammation damage via regulation of microRNA-31/MyD88[J]. Int Immunopharmacol, 2021, 101(Pt B): 108283. [15] PASSOS F, ARAÚJO-FILHO HG, MONTEIRO BS, et al. Anti-inflammatory and modulatory effects of steroidal saponins and sapogenins on cytokines: A review of pre-clinical research[J]. Phytomedicine, 2022, 96: 153842. doi: 10.1016/j.phymed.2021.153842 [16] WANG Y, CUI JQ, LIU M, et al. Schisandrin C attenuates renal damage in diabetic nephropathy by regulating macrophage polarization[J]. Am J Transl Res, 2021, 13(1): 210-222. [17] WANG EY, WANG L, DING R, et al. Astragaloside Ⅳ acts through multi-scale mechanisms to effectively reduce diabetic nephropathy[J]. Pharmacol Res, 2020, 157: 104831. doi: 10.1016/j.phrs.2020.104831 [18] HARIKRISHNAN R, DEVI G, VAN DOAN H, et al. Study on antioxidant potential, immunological response, and inflammatory cytokines induction of glycyrrhizic acid (GA) in silver carp against vibriosis[J]. Fish Shellfish Immunol, 2021, 119: 193-208. doi: 10.1016/j.fsi.2021.09.040 [19] CHEN Y, LIU QP, SHAN ZF, et al. The protective effect and mechanism of catalpol on high glucose-induced podocyte injury[J]. BMC Complement Altern Med, 2019, 19(1): 244. doi: 10.1186/s12906-019-2656-8 [20] WANG QW, DAI XX, XIANG X, et al. A natural product of acteoside ameliorate kidney injury in diabetes db/db mice and HK-2 cells via regulating NADPH/oxidase-TGF-β/Smad signaling pathway[J]. Phytother Res, 2021, 35(9): 5227-5240. doi: 10.1002/ptr.7196 [21] GNUDI L, COWARD RJM, LONG DA. Diabetic nephropathy: Perspective on novel molecular mechanisms[J]. Trends Endocrinol Metab, 2016, 27(11): 820-830. doi: 10.1016/j.tem.2016.07.002 [22] 池杨峰, 刘爽, 王浩, 等. 黄芪汤对糖尿病肾病Ⅲ期患者蛋白尿及相关炎症因子的影响[J]. 中国中西医结合肾病杂志, 2020, 21(4): 305-308. doi: 10.3969/j.issn.1009-587X.2020.04.008CHI YF, LIU S, WANG H, et al. Effect of Huangqi Decoction on proteinuria and related inflammatory factors expression of patients with diabetic kidney disease Ⅲ[J]. Chin J Integr Tradit West Nephrol, 2020, 21(4): 305-308. doi: 10.3969/j.issn.1009-587X.2020.04.008 [23] FU J, YI ZZ, CAI MC, et al. Global transcriptomic changes in glomerular endothelial cells in mice with podocyte depletion and glomerulosclerosis[J]. Cell Death Dis, 2021, 12(7): 687. doi: 10.1038/s41419-021-03951-x [24] LIANG G, SONG LT, CHEN ZL, et al. Fibroblast growth factor 1 ameliorates diabetic nephropathy by an anti-inflammatory mechanism[J]. Kidney Int, 2018, 93(1): 95-109. doi: 10.1016/j.kint.2017.05.013 [25] WU L, LIU CJ, CHANG DY, et al. Annexin A1 alleviates kidney injury by promoting the resolution of inflammation in diabetic nephropathy[J]. Kidney Int, 2021, 100(1): 107-121. doi: 10.1016/j.kint.2021.02.025 [26] CHANG TT, CHEN JW. The role of chemokines and chemokine receptors in diabetic nephropathy[J]. Int J Mol Sci, 2020, 21(9): 3172. doi: 10.3390/ijms21093172 [27] WANG YD, WU X, GENG MY, et al. CCL24 protects renal function by controlling inflammation in podocytes[J]. Dis Markers, 2021, 2021: 8837825. [28] SHANG J, WANG LY, ZHANG Y, et al. Chemerin/ChemR23 axis promotes inflammation of glomerular endothelial cells in diabetic nephropathy[J]. J Cell Mol Med, 2019, 23(5): 3417-3428. doi: 10.1111/jcmm.14237 [29] DING LH, LIU D, XU M, et al. TLR2-MyD88-NF-κB pathway is involved in tubulointerstitial inflammation caused by proteinuria[J]. Int J Biochem Cell Biol, 2015, 69: 114-120. doi: 10.1016/j.biocel.2015.10.014 [30] CHENG JH, XU X, LI YB, et al. Arctigenin ameliorates depression-like behaviors in Toxoplasma gondii-infected intermediate hosts via the TLR4/NF-κB and TNFR1/NF-κB signaling pathways[J]. Int Immunopharmacol, 2020, 82: 106302. doi: 10.1016/j.intimp.2020.106302 [31] QI MY, HE YH, CHENG Y, et al. Icariin ameliorates streptozocin-induced diabetic nephropathy through suppressing the TLR4/NF-κB signal pathway[J]. Food Funct, 2021, 12(3): 1241-1251. doi: 10.1039/D0FO02335C [32] LIU TL, ZHANG MJ, NIU HY, et al. Astragalus polysaccharide from Astragalus Melittin ameliorates inflammation via suppressing the activation of TLR-4/NF-κB p65 signal pathway and protects mice from CVB3-induced virus myocarditis[J]. Int J Biol Macromol, 2019, 126: 179-186. doi: 10.1016/j.ijbiomac.2018.12.207 [33] TIAN H, LIU ZJ, PU YW, et al. Immunomodulatory effects exerted by Poria cocos polysaccharides via TLR4/TRAF6/NF-κB signaling in vitro and in vivo[J]. Biomed Pharmacother, 2019, 112: 108709. doi: 10.1016/j.biopha.2019.108709 [34] CHOI YH. Catalpol attenuates lipopolysaccharide-induced inflammatory responses in BV2 microglia through inhibiting the TLR4-mediated NF-κB pathway[J]. Gen Physiol Biophys, 2019, 38(2): 111-122. doi: 10.4149/gpb-2018044 [35] MAO DD, TIAN XY, MAO D, et al. A polysaccharide extract from the medicinal plant Maidong inhibits the IKK-NF-κB pathway and IL-1β-induced islet inflammation and increases insulin secretion[J]. J Biol Chem, 2020, 295(36): 12573-12587. doi: 10.1074/jbc.RA120.014357 [36] YOU SB, QIAN JC, WU GJ, et al. Schizandrin B attenuates angiotensin Ⅱ induced endothelial to mesenchymal transition in vascular endothelium by suppressing NF-κB activation[J]. Phytomedicine, 2019, 62: 152955. doi: 10.1016/j.phymed.2019.152955 -





 下载:
下载: