Effect of Arisaematis Rhizoma Processed on the Contents of Lectin Protein and Calcium Oxalate Crystal as Toxic Components
-
摘要:
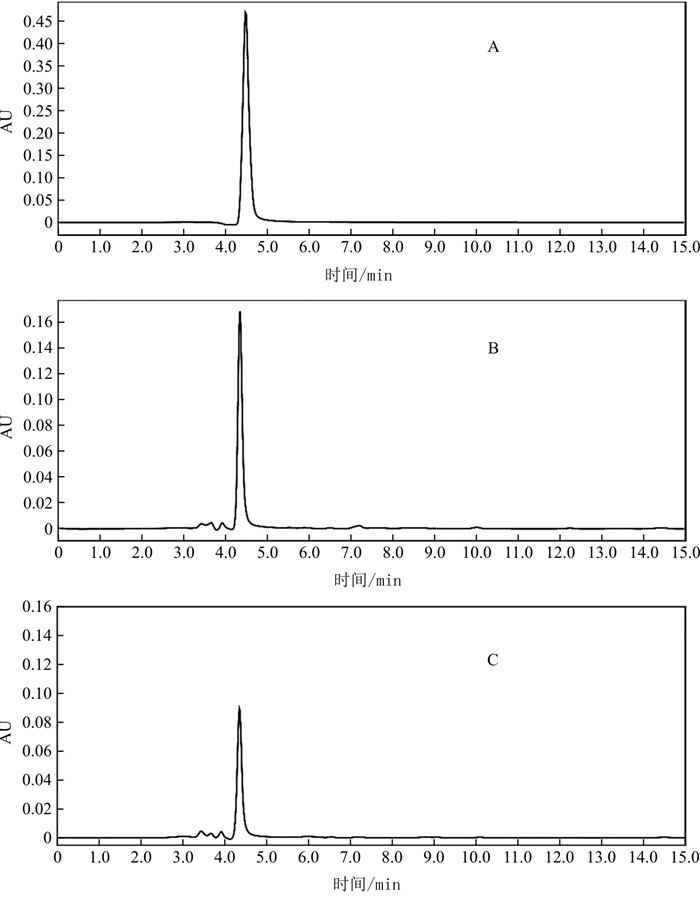
目的 探究天南星炮制前后内源性毒性成分的含量变化, 建立天南星毒性控制指标检测方法。 方法 采用HPLC法对35批天南星炮制前后草酸钙含量进行测定, 色谱柱为Megres C18(250 mm×4.6 mm, 5 μm), 流动相为0.5%磷酸水溶液, 流速0.8 mL · min-1, 检测波长220 nm, 柱温25 ℃; 采用Western blot法对32批天南星炮制前后凝集素蛋白进行检测。 结果 各批次生天南星草酸钙含量为0.78%~2.33%, 平均含量为1.43%;各批次制天南星草酸钙含量为0.29%~0.98%, 平均含量为0.60%;炮制后草酸钙含量的降幅为25.6%~78.3%。各批次天南星炮制后均未检测到凝集素蛋白条带。 结论 天南星经过复制法炮制, 可显著降低所含内源性毒性物质草酸钙及凝集素蛋白的含量。建立的草酸钙及凝集素蛋白检测方法可用于天南星毒性成分检测, 以凝集素蛋白限度作为天南星毒性控制指标更为稳定可靠, 可为天南星毒性控制指标的建立提供参考。 Abstract:OBJECTIVE The present study was aimed to investigate the effect of Arisaematis Rhizoma processed on the contents of endogenic toxic components and to establish the detection method. METHODS HPLC was applied to determinate the content of calcium oxalate. The separation was developed on Megres C18 column (250 mm×4.6 mm, 5 μm) by gradient elution with water containing 0.5% phosphoric acid as the moble phase at the flow rate of 0.8 mL·min-1, the column temperature of 25 ℃, and the detection wavelength of 220 nm. Western blot was applied to detect the content of lectin protein. RESULTS The contents of calcium oxalate of all batches of raw Arisaematis Rhizoma was 0.78%-2.33%, and average content was 1.43%;The contents of calcium oxalate of all batches of processed Arisaematis Rhizoma were 0.29%-0.98%, and average content was 0.60%; After processing, the decreasing amplitude of calcium oxalate were 25.6%-78.3%. No lectin protein was detected in all batches of processed Arisaematis Rhizoma. CONCLUSION The contents of toxic components, lectin protein and calcium oxalate, were decreased in Arisaematis Rhizoma after processed. The detection method of lectin protein and calcium oxalate can be used to detect toxic components of multi-batch Arisaematis Rhizoma, and can provide reference for the establishment of toxicity control index of Arisaematis Rhizoma. It is stable and reliable that consider the limit of lectin protein as toxicity control index of Arisaematis Rhizoma. -
Key words:
- Arisaematis Rhizoma /
- process /
- toxic components /
- calcium oxalate crystal /
- lectin protein /
- content determination
-
表 1 天南星样品信息
Table 1. Information of Arisaematis Rhizoma samples
编号 产地 批号 编号 产地 批号 编号 产地 批号 AR1 贵州铜仁 200101 AR25 四川凉山 20210324 ARP-14 自制 2020100908 AR2 四川绵阳 190201 AR26 四川江油 C216119001101 ARP-15 自制 2020100909 AR3 吉林辽源 190901 AR27 四川凉山 C216119001102 ARP-16 自制 2020100910 AR4 云南大理 C2161200021 AR28 湖南怀化 C216120001101 ARP-17 自制 2020100912 AR5 湖南怀化 C2161200011 AR29 湖南怀化 C216120001102 ARP-18 自制 2020100913 AR6 四川雅安 C2161190011 AR30 贵州铜仁 C216119002101 ARP-19 自制 2021022402 AR7 河北安平 20201001 AR31 贵州铜仁 C216119002102 ARP-20 自制 2021022403 AR8 河北安国 20201002 AR32 吉林辽源 19090101 ARP-21 自制 2021022405 AR9 贵州铜仁 20201004 AR33 四川江油 19020101 ARP-22 自制 2021022407 AR10 四川革庆 20201005 AR34 四川绵阳 19020102 ARP-23 自制 2021022408 AR11 四川雅安 20201006 AR35 贵州铜仁 20010102 ARP-24 自制 2021022409 AR12 云南富源 20201007 ARP-1 自制 2020080309 ARP-25 自制 2021022410 AR13 云南南华 20201008 ARP-2 自制 2020080310 ARP-26 自制 2021051901 AR14 云南宣威 20201009 ARP-3 自制 2020080311 ARP-27 自制 2021051902 AR15 云南楚雄 20201010 ARP-4 自制 2020080313 ARP-28 自制 2021051903 AR16 云南大理 20201011 ARP-5 自制 2020080315 ARP-29 自制 2021051904 AR17 云南曲靖 20201012 ARP-6 自制 2020080316 ARP-30 自制 2021051905 AR18 云南沾益 20201013 ARP-7 自制 2020092209 ARP-31 自制 2021051906 AR19 湖北荆州 20201102 ARP-8 自制 2020092210 ARP-32 自制 2021051907 AR20 云南迪庆 20201201 ARP-9 自制 2020092212 ARP-33 自制 2021051909 AR21 四川绵阳 20210102 ARP-10 自制 2020092213 ARP-34 自制 2021051910 AR22 云南大理 20210201 ARP-11 自制 2020092214 ARP-35 自制 2021051912 AR23 云南昭通 20210202 ARP-12 自制 2020092215 AR24 四川江油 20210310 ARP-13 自制 2020092216 注: AR.生天南星; ARP.制天南星(由对应编号生天南星制得)。 表 2 加样回收率测定(n=6)
Table 2. Results of recovery test (n=6)
编号 样品量/g 草酸含量/mg 草酸加入量/mg 实测量/mg 加样回收率/% 平均值/% RSD/% 1 0.250 1 1.300 1 1.28 2.504 8 94.12 93.62 0.59 2 0.250 3 1.301 2 1.28 2.493 5 93.15 3 0.250 2 1.300 7 1.28 2.503 0 93.94 4 0.250 2 1.300 7 1.28 2.506 6 94.21 5 0.250 3 1.301 2 1.28 2.489 9 92.87 6 0.250 2 1.300 7 1.28 2.496 6 93.44 表 3 天南星中草酸钙含量(n=2)
Table 3. Content of calcium oxalate in Arisaematis Rhizoma (n=2)
样品 含量/% 样品 含量/% 降低率/% 样品 含量/% 样品 含量/% 降低率/% AR1 1.69 ARP1 0.37 78.3 AR19 1.31 ARP19 0.43 66.9 AR2 1.36 ARP2 0.60 56.0 AR20 1.28 ARP20 0.61 51.8 AR3 1.58 ARP3 0.38 76.0 AR21 1.33 ARP21 0.29 78.1 AR4 1.37 ARP4 0.88 35.7 AR22 1.33 ARP22 0.48 63.9 AR5 1.56 ARP5 0.64 59.1 AR23 1.38 ARP23 0.53 61.6 AR6 1.08 ARP6 0.53 51.0 AR24 0.84 ARP24 0.43 49.3 AR7 1.76 ARP7 0.46 73.8 AR25 1.88 ARP25 0.54 71.1 AR8 1.74 ARP8 0.46 73.5 AR26 1.74 ARP26 0.82 53.2 AR9 1.20 ARP9 0.40 66.9 AR27 1.75 ARP27 0.80 54.6 AR10 1.18 ARP10 0.40 66.5 AR28 1.15 ARP28 0.85 25.8 AR11 1.01 ARP11 0.35 65.3 AR29 1.68 ARP29 0.69 59.2 AR12 1.28 ARP12 0.64 50.3 AR30 1.36 ARP30 0.58 57.3 AR13 1.88 ARP13 0.80 57.5 AR31 1.17 ARP31 0.67 42.6 AR14 1.69 ARP14 0.70 58.6 AR32 0.78 ARP32 0.50 35.0 AR15 1.96 ARP15 0.62 68.5 AR33 1.34 ARP33 0.65 51.5 AR16 2.33 ARP16 0.95 59.4 AR34 1.34 ARP34 0.69 48.2 AR17 0.99 ARP17 0.74 25.6 AR35 1.69 ARP35 0.98 42.4 AR18 1.13 ARP18 0.49 56.3 -
[1] 国家中医药管理局. 中华本草: 精华本[M]. 上海: 上海科学技术出版社, 1998.National Administration of Traditional Chinese Medicine. Chinese Materia Medica: Essence Edition[M]. Shanghai: Shanghai science and technology press, 1998. [2] 吴皓, 钟凌云. 天南星科有毒中药刺激性作用比较研究[J]. 中国中药杂志, 2008, 33(4): 380-384. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY200804010.htmWU H, ZHONG LY. Study on irritation of calcium oxalate crystal in Araceae plants[J]. China J Chin Mater Med, 2008, 33(4): 380-384. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY200804010.htm [3] 葛秀允, 吴皓. 天南星科有毒中药毒针晶的组成成分分析[J]. 药物分析杂志, 2010, 30(2): 190-193. https://www.cnki.com.cn/Article/CJFDTOTAL-YWFX201002003.htmGE XY, WU H. Analysis of the composition of poisonous raphides in Araceae plant[J]. Chin J Pharm Anal, 2010, 30(2): 190-193. https://www.cnki.com.cn/Article/CJFDTOTAL-YWFX201002003.htm [4] 刘先琼, 吴皓, 郁红礼, 等. 半夏、掌叶半夏及禹白附凝集素蛋白的刺激性研究[J]. 中国实验方剂学杂志, 2011, 17(18): 244-247. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201118071.htmLIU XQ, WU H, YU HL, et al. Irritability of agglutinin from Pinellia ternata breit, Pinellia pedtaisecta schott and Typhonium giganteum engl[J]. Chin J Exp Tradit Med Formulae, 2011, 17(18): 244-247. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201118071.htm [5] LIU XQ, WU H, YU HL, et al. Purification of a lectin from Arisaema erubescens (wall. ) schott and its pro-inflammatory effects[J]. Molecules, 2011, 16(11): 9480-9494. doi: 10.3390/molecules16119480 [6] 国家药典委员会. 中华人民共和国药典: 一部[M]. 北京: 中国医药科技出版社, 2020: 58.Chinese Pharmacopoeia Commission. Pharmacopoeia of People's Republic of China: Ⅰ[M]. Beijing: China medical and technology press, 2020: 58. [7] 葛秀允. 天南星科有毒中药刺激性毒性成分及矾制解毒共性机制研究[D]. 南京: 南京中医药大学, 2009.GE XY. The poisonous components and common mechanism of detoxification processing of Araceae herbal medicine[D]. Nanjing: Nanjing University of Chinese Medicine, 2009. [8] 王卫. 天南星科4种有毒中药凝集素蛋白促炎作用机制及炮制的影响[D]. 南京: 南京中医药大学, 2019.WANG W. Study on the pro-inflammatory mechanism of four lectins in Araceae and its effect by processing[D]. Nanjing: Nanjing University of Chinese Medicine, 2019. [9] 郁红礼, 王卫, 吴皓, 等. 炮制对天南星科4种有毒中药毒性成分凝集素蛋白的影响[J]. 中国中药杂志, 2019, 44(24): 5398-5404. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201924025.htmYU HL, WANG W, WU H, et al. Effect of processing on toxic components lectin from four kinds of Araceae toxic medicines[J]. China J Chin Mater Med, 2019, 44(24): 5398-5404. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201924025.htm [10] YU HL, PAN YZ, WU H, et al. The alum-processing mechanism attenuating toxicity of Araceae Pinellia ternata and Pinellia pedatisecta[J]. Arch Pharmacal Res, 2015, 38(10): 1810-1821. -





 下载:
下载: