Study on Processing Procedure of Wine-Processed Ligustri Lucidi Fructus Based on UPLC Characteristic Chromatogram and Multicomponent Content Determination
-
摘要:
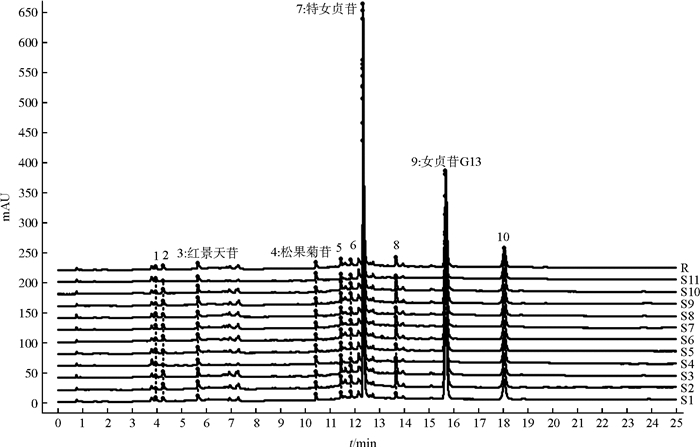
目的 建立UPLC特征图谱和多成分同时定量的分析方法, 为酒女贞子炮制过程质量评价提供依据。 方法 采用ACQUITY UPLC BEH Shield RP18(100 mm×2.1 mm, 1.7 μm)色谱柱, 以乙腈(A)-水(B)为流动相, 梯度洗脱, 流速为0.3 mL · min-1, 柱温为30 ℃, 检测波长0~3 min为284 nm, 3~25 min为224 nm, 进样量1 μL。采用化学模式识别分析方法对酒女贞子炮制过程进行质量评价。 结果 建立了女贞子炮制前后的UPLC特征图谱, 女贞子和酒女贞子分别确定了10个和14个共有峰, 指认3、4、7、9号峰为红景天苷、松果菊苷、特女贞苷、女贞苷G13, 炮制后新增成分11号峰为5-羟甲基糠醛; 聚类分析和主成分分析(PCA)将酒女贞子不同炮制过程的样品分为2类, 结合正交偏最小二乘法-判别分析(OPLS-DA)发现了导致不同炮制过程样品间差异的5个标志性化合物。定量分析了3个指标性成分, 红景天苷随着炮制时间的延长含量增加, 特女贞苷和女贞苷G13随着炮制时间的延长含量趋于稳定。 结论 所建立的UPLC特征图谱及多成分同时定量的方法准确、重复性好, 对女贞子药材及酒女贞子炮制过程的质量控制及整体性评价具有重要意义。 Abstract:OBJECTIVE Ultra Performance Liquid Chromatography (UPLC) characteristic chromatogram and simultaneous quantitative analysis method were established to provide reference for the quality evaluation of wine-processed Ligustri Lucidi Fructus with different processing procedure. METHODS ACQUITY UPLC BEH Shield RP18 (100 mm×2.1 mm, 1.7 μm) chromatographic column was used, with acetonitrile (A)-aqueous solution (B) as flow phase by gradient elution. The flow rate was 0.3 mL · min-1, column temperature was 30 ℃ and detection wavelength of 0-3 min was 284 nm, 3-25 min was 224 nm, sample injection volume was 1 μL. The quality evaluation of different processing procedure of wine-processed Ligustri Lucidi Fructus was conducted by chemical pattern recognition. RESULTS UPLC characteristic chromatogram of Ligustri Lucidi Fructus and wine-processed Ligustri Lucidi Fructus were established, identifying 10 and 14 common peaks, respectively. No. 3, 4, 7, 9 peaks were identified as Salidroside, Echinacoside, Specnuezhenide, and Ligustroflavone G13, respectively, and the new component (No. 11 peak) were identified as 5-Hydroxymethylfurfural after processing with wine. Clustering analysis and main component analysis roughly divided the wine-processed Ligustri Lucidi Fructus samples with different processing procedures into two categories. Combined with the orthogonal partial least squares-discrimination analysis, five main marker components that caused the differences between the different processing procedure samples were found. Three index components were quantitatively analyzed, while the content of Salidroside increased with prolonged processing time, and the contents of specnuezhenide and Ligustroflavone G13 stabilized with prolonged processing time. CONCLUSIONS The UPLC characteristic chromatogram and simultaneous quantitative methods established in this study are accurate and repetitive, which is of great significance for the quality control and overall evaluation of Ligustri Lucidi Fructus and the processing procedure of wine-processed Ligustri Lucidi Fructust. -
表 1 11批女贞子来源信息
Table 1. Source information of 11 batches of Ligustri Lucidi Fructus
编号 产地 编号 产地 S1 江苏徐州 S7 江苏盐城 S2 江苏徐州 S8 江苏邳州 S3 江苏徐州 S9 江苏邳州 S4 山东枣庄 S10 江苏徐州 S5 山东枣庄 S11 江苏徐州 S6 河南新乡 表 2 3个成分的线性回归方程与线性范围
Table 2. Linear regression equations with linear ranges for the three components
成分 回归方程 相关系数R2 线性范围/(μg·mL-1) 红景天苷 Y=4 797.6X-1 697.5 0.999 6 12.13~301.18 特女贞苷 Y=4 794.0X-3 785.6 0.999 2 9.88~247.76 女贞苷G13 Y=3 817.2X+25 885.8 0.999 1 10.83~271.13 表 3 3个成分的加样回收率试验结果(n=9)
Table 3. Results of additional sample recovery test for three components (n=9)
成分 称样量/g 样品含量/mg 对照品加入量/mg 测得量/mg 回收率/% RSD/% 红景天苷 0.1596
0.1551
0.1592
0.1581
0.1577
0.1583
0.1525
0.1532
0.15270.371
0.361
0.370
0.368
0.367
0.368
0.355
0.356
0.3550.200
0.200
0.200
0.400
0.400
0.400
0.600
0.600
0.6000.570
0.560
0.566
0.780
0.767
0.761
0.944
0.952
0.95199.30
99.89
98.02
103.02
100.00
98.21
98.35
99.37
99.401.51 特女贞苷 0.1501
0.1526
0.1582
0.1532
0.1549
0.1552
0.1554
0.1566
0.15274.320
4.392
4.553
4.409
4.458
4.467
4.473
4.507
4.3951.924
1.924
1.924
3.848
3.848
3.848
5.772
5.772
5.7726.264
6.334
6.459
8.152
8.230
8.161
10.270
10.181
10.075101.03
100.96
99.07
97.27
98.02
96.01
100.44
98.30
98.411.73 女贞苷G13 0.1570
0.1572
0.1568
0.1517
0.1511
0.1524
0.1596
0.1598
0.15932.832
2.836
2.828
2.736
2.726
2.749
2.879
2.883
2.8741.423
1.423
1.423
2.846
2.846
2.846
4.269
4.269
4.2694.315
4.216
4.250
5.603
5.675
5.581
7.204
7.397
7.321104.17
96.97
99.90
100.70
103.62
99.48
101.29
105.75
104.182.78 -
[1] 国家药典委员会. 中华人民共和国药典: 一部[S]. 北京: 中国医药科技出版社, 2020: 47-48.National Pharmacopoeia Commission. Pharmacopoeia of People's Republic of China: I[S]. Beijing: China medical science press, 2020: 47-48. [2] 吴皓, 李飞. 中药炮制学[M]. 2版. 北京: 人民卫生出版社, 2016: 350-351.WU H, LI F. The Processing of Chinese Materia Medica[M]. 2nd edition. Beijing: People's medical publishing house, 2016: 350-351. [3] 纪鑫, 刘晓谦, 高陆, 等. 基于女贞子和酒女贞子中主要差异性成分的体内药代动力学研究[J]. 药学学报, 2021, 56(12): 3518-3525.JI X, LIU XQ, GAO L. Pharmacokinetics of the main components differing between Ligustri Lucidi Fructus and its wine-steamed product[J]. Acta Pharmacol Sin, 2021, 56(12): 3518-3525. [4] 李佳阳, 刘峰, 林玉梅, 等. 蒸制对中药化学成分的影响研究进展[J]. 中华中医药学刊, 2022, 40(2): 128-131.LI JY, LIU F, LIN YM. Effect of Steaming on the chemical components of traditional Chinese medicine[J]. Chin Arch Tradit Chin Med, 2022, 40(2): 128-131. [5] 刘婵婵, 杨薪正, 钱卫东, 等. 中药女贞子的化学成分及药理作用研究进展[J]. 海峡药学, 2021, 33(2): 37-39. https://www.cnki.com.cn/Article/CJFDTOTAL-HAIX202102013.htmLIU CC, YANG XZ, QIAN WD. Advances in research on chemical constituents and pharmacological effects of Ligustrum lucidum[J]. Strait Pharmaceut J, 2021, 33(2): 37-39. https://www.cnki.com.cn/Article/CJFDTOTAL-HAIX202102013.htm [6] 高赛, 周欣, 陈华国. 女贞子化学成分及质量控制研究进展[J]. 中国中医药信息杂志, 2018, 25(12): 133-136. https://www.cnki.com.cn/Article/CJFDTOTAL-XXYY201812034.htmGAO S, ZHOU X, CHEN HG. Research progress in chemical components and quality control of ligustri lucidi fructus[J]. Chin J Inf Tradit Chin Med, 2018, 25(12): 133-136. https://www.cnki.com.cn/Article/CJFDTOTAL-XXYY201812034.htm [7] 杨阳, 易骏, 马利, 等. 多指标正交试验优化女贞子中环烯醚萜苷的提取工艺[J]. 中国药房, 2016, 27(28): 3970-3973. doi: 10.6039/j.issn.1001-0408.2016.28.25YANG Y, YI J, MA L, et al. Optimization of extraction process for iridoid glycosides from Ligustrum lucidum by multi-index orthogonal test[J]. China Pharm, 2016, 27(28): 3970-3973. doi: 10.6039/j.issn.1001-0408.2016.28.25 [8] 陈进芳, 翁育才. 3种不同炮制方法对女贞子中7种化学成分变化的影响[J]. 中国现代中药, 2021, 23(7): 1260-1265. https://www.cnki.com.cn/Article/CJFDTOTAL-YJXX202107017.htmCHEN JF, WENG YC. Effects of three processing methods on changes of seven chemical constituents in ligustri lucidi fructus[J]. Mod Chin Med, 2021, 23(7): 1260-1265. https://www.cnki.com.cn/Article/CJFDTOTAL-YJXX202107017.htm [9] 刘亭亭, 王萌. 女贞子化学成分与药理作用研究进展[J]. 中国实验方剂学杂志, 2014, 20(14): 228-234. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201414061.htmLIU TT, WANG M. Research progress of chemical composition and pharmacological effects of fructus ligustri lucidi[J]. Chin J Exp Tradit Med Formulae, 2014, 20(14): 228-234. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201414061.htm [10] 张明发, 沈雅琴. 女贞子及其活性成分防治呼吸系统疾病的药理作用研究进展[J]. 药物评价研究, 2020, 43(11): 2349-2362. https://www.cnki.com.cn/Article/CJFDTOTAL-YWPJ202011035.htmZHANG MF, SHEN YQ. Research progress on pharmacologic effects of Fructus Ligustri Lucidi and its active components in prophylaxis and treatment for respiratory system disease[J]. Drug Eval Res, 2020, 43(11): 2349-2362. https://www.cnki.com.cn/Article/CJFDTOTAL-YWPJ202011035.htm [11] 纪鑫, 刘晓谦, 肖苏萍, 等. 女贞子中非三萜类成分的定性定量研究[J]. 中国中药杂志, 2019, 44(8): 1615-1622. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201908015.htmJI X, LIU XQ, XIAO SP, et al. Qualitative and quantitative analysis on non-triterpenoids in Ligustri Lucidi Fructus[J]. China J Chin Mater Med, 2019, 44(8): 1615-1622. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201908015.htm [12] 姜秋, 蒋海强, 李慧芬, 等. 女贞子酒蒸过程中4种裂环环烯醚萜苷类成分的动态变化[J]. 中成药, 2014, 36(12): 2561-2564. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYA201412026.htmJIANG Q, JIANG HQ, LI HF, et al. Contents dynamic changes of four secoiridoid glycosides under steaming time spans with wine in Ligubtrum lucidum Ait[J]. Chin Tradit Pat Med, 2014, 36(12): 2561-2564. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYA201412026.htm [13] 姜秋, 蒋海强, 李慧芬, 等. 女贞子酒蒸过程中5种苯乙醇类成分的变化规律[J]. 中国实验方剂学杂志, 2014, 20(16): 60-63. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201416017.htmJIANG Q, JIANG HQ, LI HF, et al. Contents changes of five phenylethanols under steaming time spans with wine in ligustri lucidi fructus[J]. Chin J Exp Tradit Med Formulae, 2014, 20(16): 60-63. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201416017.htm [14] 杜亚朋, 王美, 李璐遥, 等. 基于化合物稳定性探讨炮制对含环烯醚萜类成分中药药性及功效影响的研究进展[J]. 中草药, 2021, 52(16): 5039-5051. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202116029.htmDU YP, WANG M, LI LY, et al. Research progress on effect of processing on properties and efficacy of traditional Chinese medicine containing iridoid terpenoids based on stability of compounds[J]. Chin Tradit Herb Drugs, 2021, 52(16): 5039-5051. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO202116029.htm [15] 张学兰, 宋梦晗, 姜秋, 等. 女贞子炮制前后环烯醚萜苷类成分转化机制研究[J]. 辽宁中医杂志, 2017, 44(12): 2602-2604. https://www.cnki.com.cn/Article/CJFDTOTAL-LNZY201712046.htmZHANG XL, SONG MH, JIANG Q, et al. Research on transformation mechanism of secoiridoid glycosides components in ligubtrum lucidum before and after processing[J]. Liaoning J Tradit Chin Med, 2017, 44(12): 2602-2604. https://www.cnki.com.cn/Article/CJFDTOTAL-LNZY201712046.htm [16] 梁晓, 吴鹏, 张学兰, 等. 女贞子不同炮制品HPLC指纹图谱色谱峰的归属与比较[J]. 中药材, 2015, 38(11): 2288-2292. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYCA201511014.htmLIANG X, WU P, ZHANG XL, et al. Identification and comparison of constituents of different processed products of ligubtrum lucidum fruit by HPLC fingerprint[J]. J Chin Med Mater, 2015, 38(11): 2288-2292. https://www.cnki.com.cn/Article/CJFDTOTAL-ZYCA201511014.htm -





 下载:
下载: