Effect of Xiangsha Liujunzi Decoction Regulating CircRNA-0067835 on Cholesterol Efflux in Hyperlipidemia Rats with Spleen Deficiency
-
摘要:
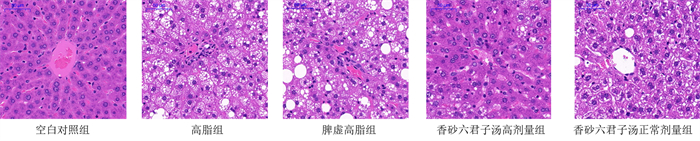
目的 探讨香砂六君子汤对脾虚高脂血症大鼠脂质沉积的影响及分子生物学机制。 方法 30只SD大鼠随机分成空白对照组、高脂组、脾虚高脂组、香砂六君子汤高剂量组及香砂六君子汤正常剂量组。使用不节饮食加游泳直至力竭的复合方法15 d, 建立脾虚模型。随后饲喂高脂饲料10周, 检测大鼠血脂水平, 确定高脂血症模型复制成功后, 香砂六君子汤高剂量组及正常剂量组分别给予香砂六君子汤11.34、5.67 g·kg-1·d-1灌胃, 其余3组给予相应体积的生理盐水灌胃, 4周后取材。检测5组大鼠血脂水平。HE染色观察肝脏病理变化。qPCR法检测CircRNA-0067835、miR-155基因水平。qPCR及Western blot法检测PPARγ、LXR、ABCA1、ABCG1 mRNA及蛋白表达水平。 结果 与空白对照组比较, 高脂组和脾虚高脂组HE染色可见明显肿胀的肝脏细胞以及清晰的脂肪空泡, 血清TC、TG、LDL-C水平以及miR-155基因表达升高(P < 0.01), HDL-C水平, CircRNA-0067835基因表达,PPARγ、LXR、ABCA1、ABCG1 mRNA和蛋白表达均降低(P < 0.05,P < 0.01)。相较于脾虚高脂组, 香砂六君子汤高剂量组及正常剂量组TC、TG和LDL-C水平显著降低(P < 0.01), HDL-C水平显著升高(P < 0.01), 肿胀的肝脏细胞得到明显改善、脂肪空泡减少, miR-155表达降低(P < 0.01), CircRNA-0067835表达升高(P < 0.01), PPARγ、LXR、ABCA1、ABCG1 mRNA及蛋白表达均升高(P < 0.05,P < 0.01)。香砂六君子汤高剂量组与正常剂量组之间部分存在统计学意义(P < 0.05, P < 0.01)。 结论 香砂六君子汤可能通过影响CircRNA-0067835调节PPARγ介导的胆固醇外排过程改善高脂血症大鼠胆固醇代谢异常, 进而起到防治血脂异常的作用。 Abstract:OBJECTIVE To investigate the effect of Xiangsha Liujunzi Decoction on lipid deposition in rats with spleen deficiency and hyperlipidemia and its molecular biological mechanism. METHODS Thirty SD rats were randomly divided into 5 groups: blank control group, high-fat group, spleen-deficiency and high-fat group, high-dose Xiangsha Liujunzi Decoction group, and normal-dose Xiangsha Liujunzi Decoction group. A compound method of unregulated diet and swimming until exhaustion was used for 15 days to establish a model of spleen deficiency. After feeding the high-fat diet for 10 weeks, the blood lipid levels of rats were tested to confirm that the hyperlipidemia model was successfully replicated. Xiangsha Liujunzi Decoction high-dose group and normal-dose group were given Xiangsha Liujunzi Decoction of 11.34, 5.67 g·kg-1·d-1 intragastrically, the other 3 groups were given corresponding volume of normal saline intragastrically, and the samples were taken 4 weeks later. The blood lipid levels of the 5 groups of rats were detected. HE staining was used to observe the pathological changes of the liver. qPCR method was used to detect CircRNA-0067835 and miR-155 gene levels. The expression levels of PPARγ, LXR, ABCA1, ABCG1 mRNA and protein were detected by qPCR and Western blot. RESULTS Compared with the blank control group, HE staining of high-fat group and spleen-deficiency and high-fat group showed obvious swollen liver cells and clear fatty vacuoles. The serum contents of TC, TG, LDL-C and the expression of miR-155 increased (P < 0.01), HDL-C level, the expression of CircRNA-0067835, the mRNA and protein expressions of PPARγ, LXR, ABCA1, ABCG1 all decreased (P < 0.05, P < 0.01). Compared with the spleen-deficiency and high-fat group, the TC, TG and LDL-C levels of Xiangsha Liujunzi Decoction high-dose group and normal-dose group significantly reduced (P < 0.01), and the HDL-C level significantly increased (P < 0.01), swollen liver cells were significantly improved, fatty vacuoles reduced, miR-155 expression decreased (P < 0.01), CircRNA-0067835 expression increased (P < 0.01), PPARγ, LXR, ABCA1, ABCG1 mRNA and protein expressions all increased (P < 0.05, P < 0.01). There was statistical significance between Xiangsha Liujunzi Decoction high-dose group and the normal-dose group (P < 0.05, P < 0.01). CONCLUSION Xiangsha Liujunzi Decoction may improve cholesterol metabolism in hyperlipidemia rats by influencing CircRNA-0067835 to regulate the cholesterol efflux process mediated by PPARγ, thereby preventing and treating dyslipidemia. -
Key words:
- hyperlipidemia /
- spleen deficiency /
- Xiangsha Liujunzi Decoction /
- cholesterol metabolism /
- CircRNA-0067835 /
- miR-155 /
- PPARγ
-
表 1 qPCR引物序列
Table 1. The qPCR primer sequence
基因 引物序列 长度/
bpCircRNA- 上游: GTTTCATACTTTACGCTTGT 734 0067835 下游: GCCTGGGTTTGTTTCT PPARγ 上游: ACCACTCGCATTCCTTTGAC 156 下游: TGATCGCACTTTGGTATTCTTG LXR 上游: AGACCAGGGAGGCAACACTT 157 下游: CTTTTGTGGACGAAGCTCTG ABCA1 上游: GAGCAAAGCCAAGCATCTTC 172 下游: TAGAACGGGCAGGTTGGTAG ABCG1 上游: CTGTGCGTTTTGTGCTGTTC 185 下游: GTAGGCTGGGATGGTGTCAA GAPDH 上游GTGTTTCCTCGTCCCGTAGA 202 下游CCTTGACTGTGCCGTTGAAT miR-155 GGCCTCCTACCTGTTAGCATTAA - U6 上游: CTCGCTTCGGCAGCACATATACT - 下游: ACGCTTCACGAATTTGCGTGTC 表 2 各组大鼠血脂水平比较(x±s, mmol·L-1, n=6)
Table 2. The comparison of serum lipid levels in each group(x±s, mmol·L-1, n=6)
TC TG LDL-C HDL-C 空白对照组 1.28±0.02 0.41±0.02 0.19±0.2 1.24±0.03 高脂组 1.65±0.02** 0.65±0.02** 0.33±0.03** 0.93±0.03** 脾虚高脂组 1.75±0.03** 0.73±0.02** 0.38±0.02** 0.80±0.06** 香砂六君子汤高剂量组 1.48±0.04▲▲△△ 0.51±0.02▲▲ 0.23±0.02▲▲△ 1.13±0.03▲▲△ 香砂六君子汤正常剂量组 1.57±0.02▲▲ 0.52±0.01▲▲ 0.27±0.02▲▲ 1.05±0.03▲▲ 注: 与空白对照组比较, **P < 0.01;与脾虚高脂组比较, ▲▲P < 0.01;与香砂六君子汤正常剂量组比较, △P < 0.05, △△P < 0.01。 -
[1] YANG WY, XIAO JZ, YANG ZJ, et al. Serum lipids and lipoproteins in Chinese men and women[J]. Circulation, 2012, 125(18): 2212-2221. doi: 10.1161/CIRCULATIONAHA.111.065904 [2] 李中梓. 医宗必读[M]. 王卫, 张艳军, 徐力, 等点校. 天津: 天津科学技术出版社, 1999: 349.LI ZZ. Required Readings from the Medical Ancestors[M]. WANG W, ZHANG YJ, XU L, et al, collated. Tianjin: Tianjin science and technology press, 1999: 349. [3] 朱美林. 脾虚状态对高脂血症大鼠肝脏脂质沉积的影响及机制研究[D]. 沈阳: 辽宁中医药大学, 2016.ZHU ML. The effect and mechanism of spleen virtual state on liver lipid deposition in rats with hyperlipidemia[D]. Shenyang: Liaoning University of Traditional Chinese Medicine, 2016. [4] 陈丝, 宋囡, 崔馨月, 等. 香砂六君子汤对脾虚高脂血症模型大鼠胆固醇逆向转运的影响[J]. 中医杂志, 2019, 60(17): 1493-1498. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYZ201917013.htmCHEN S, SONG N, CUI XY, et al. Effect of Xiangsha Liujunzi Decoction on reverse cholesterol transport of spleen deficiency and hyperlipidemia model rats[J]. J Tradit Chin Med, 2019, 60(17): 1493-1498. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZYZ201917013.htm [5] 陈丝, 贾连群, 宋囡, 等. 香砂六君子汤对脾虚高脂血症大鼠dyHDL的影响[J]. 中国实验方剂学杂志, 2019, 25(7): 22-28. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201907039.htmCHEN S, JIA LQ, SONG N, et al. Effect of Xiangsha Liu junzitang on dyHDL in hyperlipidemia rats with spleen deficiency[J]. Chin J Exp Tradit Med Formulae, 2019, 25(7): 22-28. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201907039.htm [6] MENG XY, GUO J, FANG WW, et al. Liver microRNA-291b-3p promotes hepatic lipogenesis through negative regulation of adenosine 5'-monophosphate (AMP)-activated protein kinase α1[J]. J Biol Chem, 2016, 291(20): 10625-10634. doi: 10.1074/jbc.M116.713768 [7] MATTIS AN, SONG GS, HITCHNER K, et al. A screen in mice uncovers repression of lipoprotein lipase by microRNA-29a as a mechanism for lipid distribution away from the liver[J]. Hepatology, 2015, 61(1): 141-152. doi: 10.1002/hep.27379 [8] LI XL, LIAN FZ, LIU C, et al. Isocaloric pair-fed high-carbohydrate diet induced more hepatic steatosis and inflammation than high-fat diet mediated by miR-34a/SIRT1 axis in mice[J]. Sci Rep, 2015, 5: 16774. doi: 10.1038/srep16774 [9] ZHANG CZ, CHEN KC, WEI RL, et al. The circFASN/miR-33a pathway participates in tacrolimus-induced dysregulation of hepatic triglyceride homeostasis[J]. Signal Transduct Target Ther, 2020, 5(1): 23. doi: 10.1038/s41392-020-0105-2 [10] 王晓明, 易杰, 廖世新, 等. 脾虚证动物模型的客观评估[J]. 中华中医药杂志, 2006, 21(7): 406-408. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY200607007.htmWANG XM, YI J, LIAO SX, et al. Objective evaluation on spleen deficiency syndrome animal models[J]. China J Tradit Chin Med Pharm, 2006, 21(7): 406-408. https://www.cnki.com.cn/Article/CJFDTOTAL-BXYY200607007.htm [11] 陈奇. 中药药理实验方法学[M]. 北京: 人民卫生出版社, 1994: 205-208.CHEN Q. Pharmacological Experiment Methodology of Chinese Medicine[M]. Beijing: People's health publishing house, 1994: 205-208. [12] 郎卿, 虞芳华. 303例代谢综合征中医证候特征分析[J]. 上海中医药杂志, 2010, 44(10): 27-29. https://www.cnki.com.cn/Article/CJFDTOTAL-SHZZ201010014.htmLANG Q, YU FH. Characteristics of Chinese medical pattern of metabolic syndrome in 303 patients[J]. Shanghai J Tradit Chin Med, 2010, 44(10): 27-29. https://www.cnki.com.cn/Article/CJFDTOTAL-SHZZ201010014.htm [13] WANG XP, GUO H, LI Y, et al. Interactions among genes involved in reverse cholesterol transport and in the response to environmental factors in dyslipidemia in subjects from the Xinjiang rural area[J]. PLoS ONE, 2018, 13(5): e0196042. doi: 10.1371/journal.pone.0196042 [14] HAN T, LV YG, WANG SJ, et al. PPARγ overexpression regulates cholesterol metabolism in human L02 hepatocytes[J]. J Pharmacol Sci, 2019, 139(1): 1-8. doi: 10.1016/j.jphs.2018.09.013 [15] SHARMA AM, STAELS B. Peroxisome proliferator-activated receptor gamma and adipose tissue-understanding obesity-related changes in regulation of lipid and glucose metabolism[J]. J Clin Endocrinol Metab, 2007, 92(2): 386-395. doi: 10.1210/jc.2006-1268 [16] CHEN BJ, BYRNE FL, TAKENAKA K, et al. Analysis of the circular RNA transcriptome in endometrial cancer[J]. Oncotarget, 2017, 9(5): 5786-5796. [17] ZHOU J, XIONG QM, CHEN HT, et al. Identification of the spinal expression profile of non-coding RNAs involved in neuropathic pain following spared nerve injury by sequence analysis[J]. Front Mol Neurosci, 2017, 10: 91. [18] YU GL, YANG Z, PENG TH, et al. Circular RNAs: Rising stars in lipid metabolism and lipid disorders[J]. J Cell Physiol, 2021, 236(7): 4797-4806. doi: 10.1002/jcp.30200 [19] ZHU LL, REN TT, ZHU ZX, et al. Thymosin-β4 mediates hepatic stellate cell activation by interfering with CircRNA-0067835/miR-155/FoxO3 signaling pathway[J]. Cell Physiol Biochem, 2018, 51(3): 1389-1398. doi: 10.1159/000495556 [20] KIM MJ, LEE YJ, YOON YS, et al. Apoptotic cells trigger the ABCA1/STAT6 pathway leading to PPAR-γ expression and activation in macrophages[J]. J Leukoc Biol, 2018, 103(5): 885-895. doi: 10.1002/JLB.2A0817-341RR [21] MAJDALAWIEH A, RO HS. PPARgamma1 and LXRalpha face a new regulator of macrophage cholesterol homeostasis and inflammatory responsiveness, AEBP1[J]. Nucl Recept Signal, 2010, 8: e004. [22] SCHUMACHER T, BENNDORF RA. ABC transport proteins in cardiovascular disease-A brief summary[J]. Molecules, 2017, 22(4): 589. doi: 10.3390/molecules22040589 [23] WESTERTERP M, BOCHEM AE, YVAN-CHARVET L, et al. ATP-binding cassette transporters, atherosclerosis, and inflammation[J]. Circ Res, 2014, 114(1): 157-170. doi: 10.1161/CIRCRESAHA.114.300738 [24] MIROSHNIKOVA VV, DEMINA EP, MAǏOROV NV, et al. ABCG1 transporter gene expression in peripheral blood mononuclear cells of patients with atherosclerosis[J]. Tsitologiia, 2014, 56(3): 234-240. [25] DEMINA EP, MIROSHNIKOVA VV, SCHWARZMAN AL. Role of the ABC transporters A1 and G1, key reverse cholesterol transport proteins, in atherosclerosis[J]. Mol Biol, 2016, 50(2): 223-230. -





 下载:
下载: