Study on the Dermal Delivery Effect of Hydrophilic Molecules with High Molecular Weight in Ganoderma Lucidum Fermentative Liquid by Cerosomes
-
摘要:
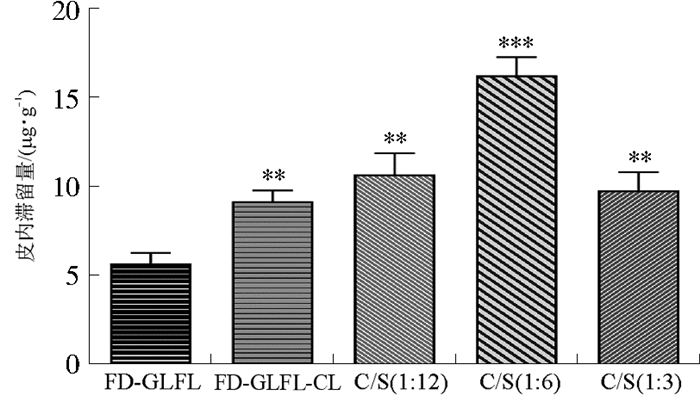
目的 研究神经酰胺脂质体(Cerosomes, CS)对灵芝发酵液(Ganoderma lucidum fermentative liquid, GLFL)大分子极性成分的经皮递送效果及机制, 为GLFL高效经皮给药制剂的开发提供依据。 方法 以水溶性大分子荧光素异硫氰酸酯-葡聚糖(Fluorescein isothiocyanate-dextran, FD)为荧光探针, 采用Franz扩散池和大鼠离体皮肤考察神经酰胺对脂质体经皮递送效果的影响, 并采用荧光显微镜观察皮内荧光分布。采用薄膜分散法制备载有灵芝发酵液的神经酰胺脂质体(GLFL-CS), 考察其形态、粒径、包封率及稳定性。应用傅里叶变换衰减全反射红外光谱法(ATR-FTIR)研究经皮渗透机制。 结果 体外经皮递送实验表明, 神经酰胺和大豆磷脂比例为1 ∶ 6的FD-GLFL-CS[C/S(1 ∶ 6)]12 h皮内滞留量最高, 分别为FD-GLFL及FD-GLFL-CL的2.87倍和1.78倍。荧光显微镜观察结果发现CS能够促进更多的药物向皮肤深层渗透。所制备的GLFL-CS呈类球形, 分布均匀, 平均粒径为(259.3±16.7)nm, GLFL蛋白多肽类成分的包封率为(43.03±0.90)%。ATR-FTIR结果表明, CS可以改变皮肤水合能力以及角质层脂质和角蛋白的主要特征峰位移及峰面积。 结论 CS可能通过增强角质层水合作用, 改变皮肤角质层结构, 降低屏障作用, 从而显著提高水溶性大分子极性物质的经皮递送。 -
关键词:
- 灵芝发酵液 /
- 荧光素异硫氰酸酯-葡聚糖 /
- 神经酰胺脂质体 /
- 经皮递送 /
- 促渗机制
Abstract:OBJECTIVE To study the dermal delivery effect and mechanism of hydrophilic molecules with high molecular weight by cerosomes, and provide foundation for the development of efficient dermal delivery system containing ganoderma lucidum fermentative liquid (GLFL). METHODS Using water-soluble macromolecule fluorescein isothiocyanate-dextran(FD) as fluorescent probe, the impact of ceramide on the dermal delivery effect of liposomes was investigated by Franz diffusion cell method and rat isolated skin, and the intradermal drug distribution was observed by fluorescence microscope. Cerasomes containing GLFL were prepared by thin-film dispersion method. The morphology, particle size, encapsulation efficiency and stability of the prepared cerasomes were measured. Attenuated total reflectance-Fourier transform infrared spectroscopy (FTIR-ATR) was applied to study the mechanism of dermal penetration enhancement mechanism. RESULTS The results of in vitro dermal delivery experiments showed that the 12-hour intradermal retention of liposomes with the ratio of ceramide to soybean phospholipid of 1 ∶ 6 was the highest, which was 2.87 times and 1.78 times of the drug solution and conventional liposomes without ceramide, respectively. Fluorescence microscopy results showed that cerosomes could promote more drugs to penetrate deeper into the skin. The prepared cerasomes were spherical in shape and uniform in size with the average particle size of (259.3±16.7)nm, and the encapsulation efficiency of protein and peptide components was (43.03±0.90)%. ATR-FTIR results showed that cerosomes could change the skin hydration capacity and the main characteristic peak displacement and peak area of stratum corneum lipids and keratins. CONCLUSION Cerasomes may enhance the stratum corneum hydration, change the structure of the skin stratum corneum, and reduce the barrier function, thereby significantly improve the dermal delivery of water-soluble polar substances with high molecular weight. -
表 1 FD-GLFL脂质体的处方
Table 1. The prescription of FD-GLFL lipidosomes
处方 磷脂膜组成/mg 大豆磷脂 胆固醇 神经酰胺3 FD-GLFL-CL 120 15 - C/S(1∶12) 120 15 10 C/S(1∶6) 120 15 20 C/S(1∶3) 120 15 40 -
[1] 王倩, 张佳婵, 王昌涛, 等. 灵芝美容护肤作用机制及其在化妆品行业中的发展现状[J]. 日用化学工业, 2019, 49(2): 118-125. doi: 10.3969/j.issn.1001-1803.2019.02.011WANG Q, ZHANG JC, WANG CT, et al. Cosmetic mechanisms of Ganoderma lucidum and its development in cosmetics[J]. China Surfactant Deterg Cosmet, 2019, 49(2): 118-125. doi: 10.3969/j.issn.1001-1803.2019.02.011 [2] YIN ZM, YANG BX, REN HW. Preventive and therapeutic effect of Ganoderma (Lingzhi) on skin diseases and care[J]. Adv Exp Med Biol, 2019, 1182: 311-321. [3] 姜晓娜, 李刚, 李翔. 灵芝在化妆品中的应用和发展趋势[J]. 中国食用菌, 2019, 38(3): 15.JIANG XN, LI G, LI X. Applications and development trends of Ganoderma lucidum in cosmetics[J]. Edible Fungi China, 2019, 38(3): 15. [4] 赵丹, 许丹妮, 王冬冬, 等. 灵芝发酵液的成分检测及美白与抗衰老功效评价[J]. 日用化学工业, 2016, 46(4): 226-230, 242.ZHAO D, XU DN, WANG DD, et al. Analysis of fermentation liquor of Ganoderma lucidum and evaluation of its whitening and anti-senility efficacy[J]. China Surfactant Deterg Cosmet, 2016, 46(4): 226-230, 242. [5] JIN YS, YUE XL, ZHANG QY, et al. Cerasomal doxorubicin with long-term storage stability and controllable sustained release[J]. Acta Biomater, 2012, 8(9): 3372-3380. doi: 10.1016/j.actbio.2012.05.022 [6] ABDELGAWAD R, NASR M, MOFTAH NH, et al. Phospholipid membrane tubulation using ceramide doping "Cerosomes": Characterization and clinical application in psoriasis treatment[J]. Eur J Pharm Sci, 2017, 101: 258-268. doi: 10.1016/j.ejps.2017.02.030 [7] 郑雪花, 于黎鹏, 杨君, 等. 氢化可的松角质类脂脂质体的理化性质及其体外经皮渗透研究[J]. 中国新药杂志, 2017, 26(15): 1822-1826.ZHENG XH, YU LP, YANG J, et al. Physicochemical properties and percutaneous permeability in vitro of hydrocortisone stratum corneum lipids liposomes[J]. Chin J New Drugs, 2017, 26(15): 1822-1826. [8] FATHI-AZARBAYJANI A, NG KX, CHAN YW, et al. Lipid vesicles for the skin delivery of diclofenac: Cerosomes vs. other lipid suspensions[J]. Adv Pharm Bull, 2015, 5(1): 25-33. [9] ABRAHAM W, DOWNING DT. Interaction between corneocytes and stratum corneum lipid liposomes in vitro[J]. Biochim Biophys Acta, 1990, 1021(2): 119-125. doi: 10.1016/0005-2736(90)90023-H [10] TOKUDOME Y, SAITO Y, SATO F, et al. Preparation and characterization of ceramide-based liposomes with high fusion activity and high membrane fluidity[J]. Colloids Surf B Biointerfaces, 2009, 73(1): 92-96. doi: 10.1016/j.colsurfb.2009.05.002 [11] SCHWARZ JC, PAGITSCH E, VALENTA C. Comparison of ATR-FTIR spectra of porcine vaginal and buccal mucosa with ear skin and penetration analysis of drug and vehicle components into pig ear[J]. Eur J Pharm Sci, 2013, 50(5): 595-600. doi: 10.1016/j.ejps.2012.12.020 [12] 王荷逸, 黄思凡, 王邦国, 等. 水溶性药物脂质体制备方法的研究进展[J]. 中国药学杂志, 2020, 55(23): 1935-1938.WANG HY, HUANG SF, WANG BG, et al. Progress on preparation method of water-soluble drug liposomes[J]. Chin Pharm J, 2020, 55(23): 1935-1938. -





 下载:
下载: