Studies on the Metabolites of Caudatin-3-O-β-D-Cymaropyranoside in Rats
-
摘要:
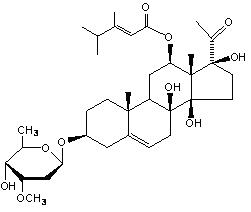
目的 研究白首乌中甾苷成分告达庭-3-O-β-D-磁麻糖苷在大鼠体内的代谢情况。 方法 大鼠单剂量灌胃给予20 mg/kg的告达庭-3-O-β-D-磁麻糖苷,分别收集24 h内的血浆、尿液、粪便和胆汁,样品经处理后,采用UPLC-Q-TOF分析,结合MetabolynxTM软件初步鉴定告达庭-3-O-β-D-磁麻糖苷在大鼠血浆、尿液、粪便和胆汁中的代谢产物。 结果 在血浆、尿液、粪便和胆汁样品中发现了告达庭-3-O-β-D-磁麻糖苷的原型、水解及去甲基化代谢物,其中尿液中有原型,血浆和胆汁中有水解产物,粪便中有去2个甲基化产物,胆汁中有去1个和去3个甲基化产物。 结论 告达庭-3-O-β-D-磁麻糖苷在大鼠体内可发生较为广泛的代谢,其中主要代谢类型有水解和去甲基化。 -
关键词:
- 白首乌 /
- 告达庭-3-O-β-D-磁麻糖苷 /
- C21甾苷 /
- 代谢
Abstract:OBJECTIVE To study the rats' metabolism of caudatin-3-O-β-D-cymarose glycosides, a kind of sterioside in Radix Cynanchi Auriculati. METHODS Rats were given the a single dose of caudatin-3-O-β-D-cymarose glycosides by intragastric administration (20 mg/kg). Then the rats' plasma, urine, feces, and bile during the following 24 hours were collected. In addition, the processed samples were analyzed by UPLC-Q-TOF with MetabolynxTM software for the preliminary measurement of the metabolites of caudatin-3-O-β-D-cymarose glycosides in rats' plasma, urine, feces, and bile. RESULTS The original, hydrolytic and demethylated metabolites of caudatin-3-O-β-D-cymarose glycosides were found in plasma, urine, feces, and bile samples, specifically, the original caudatin-3-O-β-D-cymarose glycosides in urine, hydrolytic metabolites in plasma and bile, two methylated metabolites in feces, along with one methylated and three demethylated metabolites in bile. CONCLUSION The metabolism of caudatin-3-O-β-D-cymarose glycosides in rats is extensive, and the main types of metabolism are hydrolysis and demethylation. -
表 1 Metabolite List窗口设置的可能的Ⅰ相、Ⅱ相代谢途径
Phase 代谢转化 质量数变化/Da 分子式变化 Ⅰ 原型 0 还原 2.015 7 +H2 脱甲基后羧基化 29.974 2 -CH2+CO2 脱甲基 -14.015 7 -CH2 脱氧 -15.994 9 -O 醇脱水 -18.010 6 -H2O 脱乙基 -28.031 3 -C2H4 脱乙酰基 -42.010 6 -C2H2O 脱3个甲基 -42.047 0 -3CH2 脱羧基 -44.997 7 -COOH 脱4个甲基 -56.062 6 -4CH2 脱乙酰基+脱羟基 -58.005 5 -C2H2O2 脱5个甲基 -70.078 3 -5CH2 脱6个甲基 -84.093 9 -6CH2 脱3,4-二甲基戊-2-酰基 -110.073 2 -C7H10O 脱D-磁麻糖基 -144.078 6 -C7H12O3 脱3,4-二甲基戊-2-酰基+脱D-磁麻糖基 -254.151 8 -C7H10O-C7H12O3 脱3,4-二甲基戊-2-酰基+脱D-磁麻糖基+ 脱乙酰基+脱羟基 -312.153 7 -C7H10O-C7H12O3- C2H2O2 脱3,4-二甲基戊-2-酰基+ 脱D-磁麻糖基+脱2个乙酰基+脱羟基 -354.167 9 -C7H10O-C7H12O3- C2H2O2-C2H2O Ⅱ 甲基化 14.015 7 +CH2 乙酰化 42.010 6 +C2H2O 硫酸化 79.956 8 +SO3 2×硫酸化 159.913 6 +2SO3 葡萄糖醛酸结合 176.032 1 +C6H8O6 3×硫酸化 239.870 4 +3SO3 硫酸化+葡萄糖醛酸结合 255.988 9 +SO3+C6H8O6 2×葡萄糖醛酸结合 352.064 2 +2C6H8O6 3×葡萄糖醛酸结合 528.096 3 +3C6H8O6 Ⅰ+Ⅱ 3,4-二甲基戊-2-酰基+D-磁麻糖结合+ 葡萄糖醛酸结合+硫酸化反应 1.837 1 -C7H10O-C7H12O3+C6H8O6+SO3 磁麻糖和葡萄糖醛酸结合反应 31.953 4 -C7H12O3+C6H8O6 3,4-二甲基戊-2-酰基结合和葡萄糖醛酸结合反应 65.958 9 -C7H10O+C6H8O6 3,4-二甲基戊-2-酰基结合+磁麻糖结合+2个葡萄糖醛酸结合反应 97.912 4 -C7H10O-C7H12O3+2C6H8O6 3,4-二甲基戊-2-酰基结合和硫酸化反应 -30.116 3 -C7H10O+SO3 3,4-二甲基戊-2-酰基结合+磁麻糖结合+2个硫酸化反应 -94.238 2 -C7H10O-C7H12O3+2SO3 表 2 CDMC及其代谢物MetabolynxTM软件处理结果
化合物 代谢产物 分子式 质量差 分子量 m/z mDa ppm tR/min 峰面积 M1 A: 原型 C35H54O10 -0.013 2 634.371 7 633.355 2 -8.6 -13.6 12.66 252.2 M2 A-C7H10O C28H44O9 -110.070 4 524.298 5 523.293 5 2.8 5.3 7.66 2.7 M3 A-2CH2 C33H50O10 -28.034 2 606.340 4 605.329 7 -2.9 -4.8 8.33 51.3 M4 A-CH2 C34H52O10 -14.018 9 620.356 0 619.345 0 -3.2 -5.2 9.78 23.4 M5 A-3CH2 C32H48O10 -42.055 4 592.324 7 591.308 5 -8.4 -14.3 10.98 100.4 -
[1] 印鑫, 丁永芳, 邵久针, 等. 白首乌的研究进展[J]. 中草药, 2019, 50(4): 992-1000. https://www.cnki.com.cn/Article/CJFDTOTAL-ZCYO201904030.htm [2] DING YF, PENG ZX, DING L, et al. Baishouwu extract suppresses the development of hepatocellular carcinoma via TLR4/MyD88/NF-κB pathway[J]. Front Pharmacol, 2019, 10: 389. doi: 10.3389/fphar.2019.00389 [3] 伍振辉, 孟娴, 丁永芳, 等. 白首乌C-21甾体总苷对人肝癌HepG2细胞增殖、侵袭与凋亡的影响[J]. 中国中药杂志, 2018, 43(9): 1915-1921. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGZY201809027.htm [4] 王冬艳, 张洪泉, 李心. 白首乌C21甾体苷诱导肝癌细胞凋亡的作用及其机制[J]. 药学学报, 2007, 42(4): 366-370. doi: 10.3321/j.issn:0513-4870.2007.04.004 [5] WANG H, WANG Q, SRIVASTAVA RK, et al. Effects of total glycosides from Baishouwu on human breast and prostate cancer cell proliferation[J]. Altern Ther Health Med, 2003, 9(5): 62-66. [6] 张如松, 叶益萍, 刘雪莉. 白首乌甾体总苷的体外抗肿瘤作用[J]. 中草药, 2000, 31(8): 599-601. doi: 10.3321/j.issn:0253-2670.2000.08.024 [7] 张如松, 叶益萍, 沈月毛, 等. 白首乌体外抑制肿瘤细胞的成分研究[J]. 药学学报, 2000, 35(6): 431-437. doi: 10.3321/j.issn:0513-4870.2000.06.008 [8] ZHANG RS, YE YP, SHEN YM, et al. Two new cytotoxic C-21 steroidal glycosides from the root of Cynanchum auriculatum[J]. Tetrahedron, 2000, 56(24): 3875-3879. doi: 10.1016/S0040-4020(00)00262-3 [9] 张建烽, 李友宾, 钱士辉, 等. 白首乌化学成分研究[J]. 中国中药杂志, 2006, 31(10): 814-816. doi: 10.3321/j.issn:1001-5302.2006.10.009 [10] 陈纪军, 张壮鑫, 周俊. 白首乌甙A、B和C的结构[J]. 云南植物研究, 1990, 12(2): 197-210. https://www.cnki.com.cn/Article/CJFDTOTAL-YOKE199002012.htm [11] PENG YR, LI YB, LIU XD, et al. Antitumor activity of C-21 steroidal glycosides from Cynanchum auriculatum Royle ex Wight[J]. Phytomedicine, 2008, 15(11): 1016-1020. doi: 10.1016/j.phymed.2008.02.021 [12] 李友宾, 彭蕴茹, 钱士辉, 等. 告达庭-3-O-β-D-磁麻糖苷作为治疗肿瘤药物的用途: 200710020032.8[P]. 2009-01-14. [13] PENG YR, LI YB, WANG DW, et al. Determination of caudatin-2, 6-dideoxy-3-O-methy-β-D-cymaropyranoside in rat plasma using liquid chromatography-tandem mass spectrometry[J]. Biomed Chromatogr, 2008, 22(6): 575-580. doi: 10.1002/bmc.971 [14] HONG C, YANG P, LI SP, et al. In vitro / in vivo metabolism of ginsenoside Rg5 in rat using ultra-performance liquid chromatography/quadrupole-time-of-flight mass spectrometry[J]. Molecules, 2018, 23(9): E2113. doi: 10.3390/molecules23092113 [15] 宋玮, 郑伟, 张洁, 等. 中药皂苷类成分的体内代谢研究进展[J]. 药学学报, 2018, 53(10): 1609-1619. https://www.cnki.com.cn/Article/CJFDTOTAL-YXXB201810004.htm [16] ZHANG W, PENG YR, DING YF. Biotransformation and metabolic profile of caudatin-2, 6-dideoxy-3-O-methy-β-d-cymaropyranoside with human intestinal microflora by liquid chromatography quadrupole time-of-flight mass spectrometry[J]. Biomed Chromatogr, 2015, 29(11): 1715-1723. doi: 10.1002/bmc.3484 [17] PENG YR, LI YB, LIU XD, et al. Apoptosis induced by caudatin in human hepatoma cell line SMMC7721[J]. Chin J Nat Med, 2008, 6(3): 210-213. doi: 10.3724/SP.J.1009.2008.00210 [18] FEI HR, CHEN HL, XIAO T, et al. Caudatin induces cell cycle arrest and caspase-dependent apoptosis in HepG2 cell[J]. Mol Biol Rep, 2012, 39(1): 131-138. doi: 10.1007/s11033-011-0721-6 [19] 梁起栋, 何明珍, 欧阳辉, 等. 超高效液相色谱-质谱联用法分析α-常春藤皂苷在大鼠体内的代谢产物[J]. 中草药, 2014, 45(13): 1883-1888. doi: 10.7501/j.issn.0253-2670.2014.13.014 [20] 刘亚丽, 宋永贵, 魏韶锋, 等. UPLC-ESI-Q-TOF-MS/MS分析白头翁皂苷B3在大鼠肠道菌群中的代谢产物[J]. 中国实验方剂学杂志, 2015, 21(23): 72-76. https://www.cnki.com.cn/Article/CJFDTOTAL-ZSFX201523019.htm [21] 曹伟宇, 冯斌, 王晓娟. 肠道菌群/肝药酶系对天然皂苷类成分的代谢研究进展[J]. 中国药房, 2016, 27(28): 3999-4002. doi: 10.6039/j.issn.1001-0408.2016.28.34 -





 下载:
下载: