Honokiol Loaded Mixed Micelles Fororal Delivery Using Novel F127 and TPGS as Carriers
doi: 10.14148/j.issn.1672-0482.2021.0376
-
摘要:
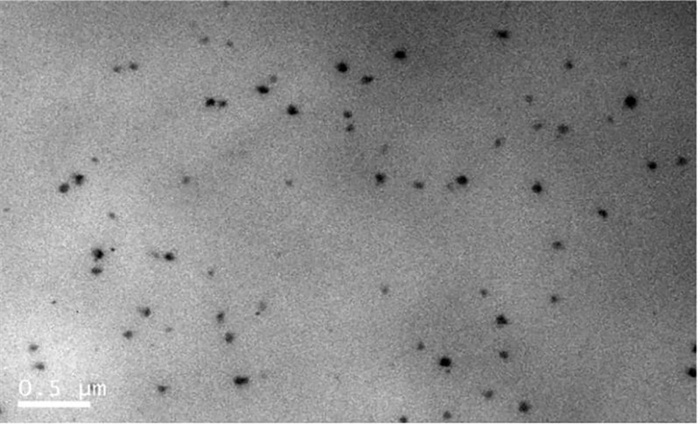
目的 旨在使用普朗尼克F127(F127)和维生素E聚乙二醇1000琥珀酸酯(TPGS)制备自组装胶束,提高和厚朴酚(HK)的口服生物利用度和抗肿瘤活性。 方法 负载和厚朴酚F127/TPGS二元混合胶束(HK-M)最佳处方采用乙醇溶剂蒸发法制备,以透射电镜(TEM)、HPLC对其进行表征,用透析袋法测定HK-M中HK的累积释放量,用Caco-2测定HK-M的渗透性,并评价其生物利用度和体内抗肿瘤活性。 结果 当F127∶TPGS(4∶1),HK-M为透明无色,粒径(23.28±2.01)nm,胶束呈球形且均匀。HK-M中HK的溶解度显著增加至4.76 mg/mL,HK-M具有较好的稳定性。HK包封于混合胶束中,可实现HK的持续释放。HK-M增强HK在Caco-2细胞单层模型中的渗透性。与游离HK相比,HK-M的相对口服生物利用度增加了1.17倍。此外,HK-M对肿瘤体积的抑制率(35.17%)高于HK组(14.86%)。 结论 HK-M能改善HK的溶解性、口服生物利用度和抗肿瘤活性。 Abstract: OBJECTIVE The purpose of this research was to develop a self-assembled micelle using biocompatible copolymers Pluronic F127 (F127) and Vitamin E d-α-Tocopheryl polyethylene glycol 1000 succinate (TPGS) to enhance the oral bioavailability and anti-cancer efficiency of honokiol (HK). METHODS The optimized prescription honokiol micelle (HK-M) was prepared by an ethanol solvent evaporation method. HK-M was characterized by transmission electron microscopy(TEM) and HPLC. The dialysis bag method was used to assesse the cumulative amount of HK released from the HK-M. Caco-2 cells were applied to measure the permeability of HK-M. The bioavailability and in vivo anti-tumor effect were also evaluated. RESULTS At the ratio of 4∶1 (F127∶TPGS), the HK-M was transparent and colourless with a small size (23.28 ± 2.01)nm and a spherical shape. The apparent solubility of HK in HK-M was dramatically increased to 4.76 mg/mL, suggesting that HK-M had good stability. Furthermore, encapsulation in micelles led to a sustained release of HK. HK-M enhances HK's permeability across Caco-2 cell monolayer. Compared with free HK, there was a 1.17-fold increase in the relative oral bioavailability for HK-M. Moreover, HK-M achieved a higher inhibition rate on tumor volume (35.17%) than HK group (14.86%). CONCLUSION-
Key words:
- honokiol /
- F127 /
- TPGS /
- micelles /
- Caco-2 cell /
- oral bioavailability /
- anti-tumor
-
Table 1. Characteristics of HK-M (x±s, n=3)
F127∶TPGS/ (mg·mL-1) Average size/nm PDI Zeta potential/mV EE/% DL/% Solubility/ (mg·mL-1) 40∶10 23.28±2.01 0.068±0.012 -2.43±0.02 91.64±0.53 8.27±0.53 4.76±0.15 Table 2. Permeability and efflux ratio of HK and HK-M in Caco-2 cell model (x±s, n=3)
Compound (50 μg·mL-1) Papp×10-7/(cm·s-1) Efflux ratio AP-BL BL-AP HK 3.16±0.35 2.57±0.29 0.81±0.01 HK-M 5.72±0.39* 4.33±0.31* 0.76±0.01 Note: Compared with HK group, *P < 0.05。 Table 3. Pharmacokinetic parameters of HK and HK-M (x±s, n=6)
Parameters HK HK-M Cmax(μg/mL) 0.415±0.023 0.940±0.075** Tmax(h) 0.958±0.102 0.708±0.102** AUC0-t (μg /mL·h) 2.396±0.303 5.211±0.501** AUC0-∞ (μg /mL·h) 3.365±0.425 5.915±0.687** t1/2(h) 5.123±1.183 3.619±0.331* MRT0-∞(h) 7.824±1.352 5.887±0.371* Note: *P < 0.05, **P < 0.01。 -
[1] PRASAD R, KATIYAR SK. Honokiol, an active compound of Magnolia plant, inhibits growth, and progression of cancers of different organs[J]. Adv Exp Med Biol, 2016, 928: 245-265. doi: 10.1007/978-3-319-41334-1_11 [2] LEE YJ, LEE YM, LEE CK, et al. Therapeutic applications of compounds in the Magnolia family[J]. Pharmacol Ther, 2011, 130(2): 157-176. doi: 10.1016/j.pharmthera.2011.01.010 [3] GOWDA AS, SUO ZC, SPRATT TE. Honokiol inhibits DNA polymerases β and λ and increases bleomycin sensitivity of human cancer cells[J]. Chem Res Toxicol, 2017, 30(2): 715-725. doi: 10.1021/acs.chemrestox.6b00451 [4] SENGUPTA S, NAGALINGAM A, MUNIRAJ N, et al. Activation of tumor suppressor LKB1 by honokiol abrogates cancer stem-like phenotype in breast cancer via inhibition of oncogenic Stat3[J]. Oncogene, 2017, 36(41): 5709-5721. doi: 10.1038/onc.2017.164 [5] PAN J, LEE Y, WANG YA, et al. Honokiol targets mitochondria to halt cancer progression and metastasis[J]. Mol Nutr Food Res, 2016, 60(6): 1383-1395. doi: 10.1002/mnfr.201501007 [6] DENG F, HU WJ, CHEN HL, et al. Development of a chitosan-based nanoparticle formulation for ophthalmic delivery of honokiol[J]. Curr Drug Deliv, 2018, 15(4): 594-600. doi: 10.2174/1567201814666170419113933 [7] DESHMANE S, DESHMANE S, SHELKE S, et al. Enhancement of solubility and bioavailability of ambrisentan by solid dispersion using Daucus carota as a drug carrier: formulation, characterization, in vitro, and in vivo study[J]. Drug Dev Ind Pharm, 2018, 44: 1001-1011. doi: 10.1080/03639045.2018.1428339 [8] SALMANI JM, LYU H, ASGHAR S, et al. Amorphous solid dispersion with increased gastric solubility in tandem with oral disintegrating tablets: A successful approach to improve the bioavailability of atorvastatin[J]. Pharm Dev Technol, 2015, 20: 465-472. doi: 10.3109/10837450.2014.882938 [9] WANG WQ, CUI CC, LI MY, et al. Study of a novel disintegrable oleanolic acid-polyvinylpolypyrrolidone solid dispersion[J]. Drug Dev Ind Pharm, 2017, 43(7): 1178-1185. doi: 10.1080/03639045.2017.1301950 [10] DENG J, ZHANG Z, LIU C, et al. The studies of N-Octyl-N-Arginine-Chitosan coated liposome as an oral delivery system of Cyclosporine A[J]. J Pharm Pharmacol, 2015, 67: 1363-1370. doi: 10.1111/jphp.12448 [11] ARAFAT M, KIRCHHOEFER C, MIKOV M, et al. Nanosized liposomes containing bile salt: A vesicular nanocarrier for enhancing oral bioavailability of BCS class Ⅲ Drug[J]. J Pharm Pharm Sci, 2017, 20: 305-318. doi: 10.18433/J3CK88 [12] WANG M, LIU M, XIE T, et al. Chitosan-modified cholesterol-free liposomes for improving the oral bioavailability of progesterone[J]. Colloids Surf B Biointerfaces, 2017, 159: 580-585. doi: 10.1016/j.colsurfb.2017.08.028 [13] FARES AR, ELMESHAD AN, KASSEM MAA. Enhancement of dissolution and oral bioavailability of lacidipine via pluronic P123/F127 mixed polymeric micelles: Formulation, optimization using central composite design and in vivo bioavailability study[J]. Drug Deliv, 2018, 25(1): 132-142. doi: 10.1080/10717544.2017.1419512 [14] HALLAN SS, KAUR P, KAUR V, et al. Lipid polymer hybrid as emerging tool in nanocarriers for oral drug delivery[J]. Artif Cells Nanomed Biotechnol, 2016, 44: 334-349. doi: 10.3109/21691401.2014.951721 [15] TANG L, FU LL, ZHU ZF, et al. Modified mixed nanomicelles with collagen peptides enhanced oral absorption of Cucurbitacin B: Preparation and evaluation[J]. Drug Deliv, 2018, 25(1): 862-871. doi: 10.1080/10717544.2018.1425773 [16] ZHANG ZH, WANG XP, AYMAN WY, et al. Studies on lactoferrin nanoparticles of gambogic acid for oral delivery[J]. Drug Deliv, 2013, 20: 86-93. doi: 10.3109/10717544.2013.766781 [17] MA CL, PAN PJ, SHAN GR, et al. Core-shell structure, biodegradation, and drug release behavior of poly(lactic acid)/poly(ethylene glycol) block copolymer micelles tuned by macromolecular stereostructure[J]. Langmuir, 2015, 31(4): 1527-1536. doi: 10.1021/la503869d [18] FARES AR, ELMESHAD AN, KASSEM MAA. Enhancement of dissolution and oral bioavailability of lacidipine via pluronic P123/F127 mixed polymeric micelles: Formulation, optimization using central composite design and in vivo bioavailability study[J]. Drug Deliv, 2018, 25(1): 132-142. doi: 10.1080/10717544.2017.1419512 [19] HOU J, WANG J, SUN E, et al. Preparation and evaluation of icariside Ⅱ-loaded binary mixed micelles using Solutol HS15 and Pluronic F127 as carriers[J]. Drug Deliv, 2016, 23(9): 3248-3256. doi: 10.3109/10717544.2016.1167270 [20] ZHANG ZH, CHEN Y, DENG J, et al. Solid dispersion of berberine-phospholipid complex/TPGS 1000/SiO2: Preparation, characterization and in vivo studies[J]. Int J Pharm, 2014, 465(1/2): 306-316. http://www.onacademic.com/detail/journal_1000036238726610_806e.html [21] ZHAO J, XU Y, WANG C, et al. Soluplus/TPGS mixed micelles for dioscin delivery in cancer therapy[J]. Drug Dev Ind Pharm, 2017, 43: 1197-1204. doi: 10.1080/03639045.2017.1304956 [22] ZHAO DJ, ZHANG HY, YANG SF, et al. Redox-sensitive mPEG-SS-PTX/TPGS mixed micelles: An efficient drug delivery system for overcoming multidrug resistance[J]. Int J Pharm, 2016, 515(1/2): 281-292. http://www.onacademic.com/detail/journal_1000039666745410_41ef.html [23] ZHANG J, FANG X, LI Z, et al. Redox-sensitive micelles composed of disulfide-linked Pluronic-linoleic acid for enhanced anticancer efficiency of brusatol[J]. Int J Nanomedicine, 2018, 13: 939-956. doi: 10.2147/IJN.S130696 [24] FARES AR, ELMESHAD AN, KASSEM MAA. Enhancement of dissolution and oral bioavailability of lacidipine via pluronic P123/F127 mixed polymeric micelles: Formulation, optimization using central composite design and in vivo bioavailability study[J]. Drug Deliv, 2018, 25(1): 132-142. doi: 10.1080/10717544.2017.1419512 [25] BRANCA C, KHOUZAMI K, WANDERLINGH U, et al. Effect of intercalated chitosan/clay nanostructures on concentrated pluronic F127 solution: A FTIR-ATR, DSC and rheological study[J]. J Colloid Interface Sci, 2018, 517: 221-229. doi: 10.1016/j.jcis.2018.02.004 [26] GUAN YB, HUANG JG, ZUO L, et al. Effect of pluronic P123 and F127 block copolymer on P-glycoprotein transport and CYP3A metabolism[J]. Arch Pharm Res, 2011, 34(10): 1719-1728. doi: 10.1007/s12272-011-1016-0 [27] WEI Z, YUAN S, HAO J, et al. Mechanism of inhibition of P-glycoprotein mediated efflux by Pluronic P123/F127 block copolymers: relationship between copolymer concentration and inhibitory activity[J]. Eur J Pharm Biopharm, 2013, 83: 266-274. doi: 10.1016/j.ejpb.2012.09.014 [28] PAWAR A, SINGH S, RAJALAKSHMI S, et al. Development of fisetin-loaded folate functionalized pluronic micelles for breast cancer targeting[J]. Artif Cells Nanomed Biotechnol, 2018, 46(S1): 347-361. doi: 10.1080/21691401.2018.1423991 [29] CHEN CH, CHIANG CJ, WU LC, et al. In vitro penetration and in vivo distribution of honokiol into the intervertebral disc in rat[J]. Anal Sci, 2015, 31(12): 1297-1302. doi: 10.2116/analsci.31.1297 [30] LI X, HOU X, DING W, et al. Sirolimus-loaded polymeric micelles with honokiol for oral delivery[J]. J Pharm Pharmacol, 2015, 67: 1663-1672. doi: 10.1111/jphp.12482 [31] KANADE R, BOCHE M, POKHARKAR V. Self-assembling raloxifene loaded mixed micelles: Formulation optimization, in vitro cytotoxicity and in vivo pharmacokinetics[J]. AAPS Pharm Sci Tech, 2018, 19(3): 1105-1115. doi: 10.1208/s12249-017-0919-6 [32] ZHANG ZH, LYU H, JIA XB, et al. Influence of vitamin E tocopherol polyethylene glycol succinate 1 000 on intestinal absorption of icariside Ⅱ[J]. Pharmazie, 2012, 67(1): 59-62. http://www.ncbi.nlm.nih.gov/pubmed/22393832 [33] LIU Y, WANG D, YANG G, et al. Comparative pharmacokinetics and brain distribution of magnolol and honokiol after oral administration of Magnolia officinalis cortex extract and its compatibility with other herbal medicines in Zhi-Zi-Hou-Po Decoction to rats[J]. Biomed Chromatogr, 2016, 30: 369-375. doi: 10.1002/bmc.3557 [34] GODUGU C, DODDAPANENI R, SINGH M. Honokiol nanomicellar formulation produced increased oral bioavailability and anticancer effects in triple negative breast cancer (TNBC)[J]. Colloids Surf B Biointerfaces, 2017, 153: 208-219. doi: 10.1016/j.colsurfb.2017.01.038 [35] WANG XH, CAI LL, ZHANG XY, et al. Improved solubility and pharmacokinetics of PEGylated liposomal honokiol and human plasma protein binding ability of honokiol[J]. Int J Pharm, 2011, 410(1/2): 169-174. http://www.onacademic.com/detail/journal_1000034021473310_66d8.html -





 下载:
下载: